Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$1c{m^3}$ of water at its boiling point absorbs $540$ calories of heat to become steam with a volume of $1671c{m^3}$.If the atmospheric pressure = $1.013 \times {10^5}N/{m^2}$ and the mechanical equivalent of heat = $4.19J/calorie$, the energy spent in this process in overcoming intermolecular forces is ..... $cal$View Solution
- 2A cylindrical tube of uniform cross-sectional area $A$ is fitted with two air tight frictionless pistons. The pistons are connected to each other by a metallic wire. Initially the pressure of the gas is $P_0$ and temperature is $T_0$, atmospheric pressure is also $P_0$. Now the temperature of the gas is increased to $2T_0$, the tension in the wire will beView Solution
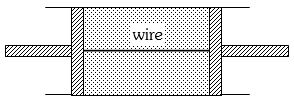
- 3View SolutionOut of the following which quantity does not depend on path
- 4In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 \,J$ of heat when $8 \,J$ of work was done on the gas. If the initial internal energy of the gas was $30 \,J$, then the final internal energy will be ........ $J$View Solution
- 5A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 6In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $30$ joules of heat and $10$ joules of work was done on the gas. If the initial internal energy of the gas was $30$ joules, then the final internal energy will be ........ $J$View Solution
- 7The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
- 8Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 9A certain amount of gas is taken through a cyclic process $(A\,B\,C\,D\,A)$ that has two isobars, one isochore and one isothermal. The cycle can be represented on a $P-V$ indicator diagram asView Solution
- 10An ideal monoatomic gas expands to twice its volume. If the process is isothermal, the magnitude of work done by the gas is $W_i$. If the process is adiabatic, the magnitude of work done by the gas is $W_a$. Which of the following is true?View Solution
