An ideal gas is taken reversibly around the cycle $a-b-c-d-a$ as shown on the temperature $T$ - entropy $S$ diagram. The most appropriate representation of above cycle on a internal energy $U$ - volume $V$ diagram is
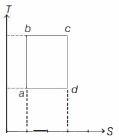

KVPY 2016, Medium
(a) In given cycle
$b c$ and $a d$ are isothermal processes $(T$ and $U$ are constants). $a b$ and $c d$ are isentropic processes ( $S=$ constant).
So, plot of above cycle on $U-V$ diagram is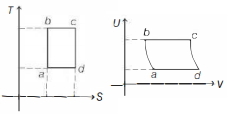
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
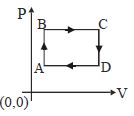
- 1Figure below shows two paths that may be taken by a gas to go from a state $A$ to a state $C.$ In process $AB,$ $400 \,J$ of heat is added to the system and in process $BC,$ $100\, J$ of heat is added to the system. The heat absorbed by the system in the process $AC$ will be ...... $J$View Solution
- 2In a certain thermodynamical process, the pressure of a gas depends on its volume as $kV ^{3}$. The work done when the temperature changes from $100^{\circ} C$ to $300^{\circ} C$ will be .......... $nR$, where $n$ denotes number of moles of a gas.View Solution
- 3View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
- 4A sample of $1$ mole gas at temperature $\mathrm{T}$ is adiabatically expanded to double its volume. If adiabatic constant for the gas is $\gamma=\frac{3}{2}$, then the work done by the gas in the process is:View Solution
- 5The figure shows $P-V$ diagram of a thermodynamic cycle. Which corresponding curve is correct?View Solution
- 6Choose the incorrect statement from the followingView Solution
$S1:$ The efficiency of a heat engine can be $1,$ but the coefficient of performance of a refrigerator can never be infinity.
$S2:$ The first law of thermodynamics is basically the principle of conservation of energy.
$S3:$ The second law of thermodynamics does not allow several phenomena consistent with the first law.
$S4:$ A process,whose only result is to transfer heat from a colder to a hotter object, is impossible.
- 7An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1 g/sec$. The calorific value of the fuel is $2 kcal/g$. Is the claim of the engineerView Solution
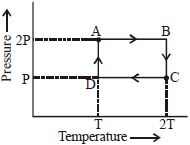
- 8An ideal monoatomic gas is taken through the thermodynamic states $A \to B \to C \to D$ via the paths shown in the figure. If $U_A, U_B, U_C$ and $U_D$ represent the internal energy of the gas in state $A, B\, C$ and $D$ respectively, then which of the following is not true?View Solution
- 9A sample of an ideal gas is contained in a cylinder. The volume of the gas is suddenly decreased. A student makes the following statements to explain the change in pressure of the gasView Solution
$I.$ The average kinetic energy of the gas atoms increases
$II.$ The atoms of the gas hit the walls of the cylinder more frequently
$III.$ Temperature of the gas remains unchanged
Which of these statements is true? - 10Two moles of an ideal monoatomic gas occupies a volume $V$ at $27^o C$. The gas expands adiabatically to a volume $2\ V$. Calculate $(a)$ the final temperature of the gas and $(b)$ change in its internal energy.View Solution
