An object is placed in a medium of refractive index $3$. An electromagnetic wave of intensity $6 \times 10^8 \mathrm{~W} / \mathrm{m}^2$ falls normally on the object and it is absorbed completely. The radiation pressure on the object would be (speed of light in free space $=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ):
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A cylinder of fixed capacity $44.8$ litre. contains a monatomic gas at standard temperature and pressure. The amount of heat required to cylinder by $10^o C$ will be. ($R=$ universal gas constant)View Solution
- 2View SolutionVariation of atmospheric pressure, with height from earth is ................
- 3Consider a gas with density $\rho $ and $\bar c$ as the root mean square velocity of its molecules contained in a volume. If the system moves as whole with velocity $v,$ then the pressure exerted by the gas isView Solution
- 4$40$ calories of heat is needed to raise the temperature of $1\, mole$ of an ideal monoatomic gas from $20°C$ to $30°C$ at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume $(R = 2\,calorie\,mol{e^{ - 1}}{K^{ - 1}})$ is ..... $calorie$View Solution
- 5The internal energy of $10\,g$ of nitrogen at $S.T.P$, is about ......... $J$View Solution
- 6One mole of an ideal gas $\left( {\frac{{{C_P}}}{{{C_V}}}\, = \gamma } \right)$ heated by law $P=\alpha V$ where $P$ is pressure of gas, $V$ is volume, $\alpha$ is a constant what is the heat capacity of gas in the process-View Solution
- 7One mole of monoatomic gas and three moles of diatomic gas are put together in a container. The molar specific heat (in $J\,{K^{ - 1}}\,mo{l^{ - 1}})$ at constant volume is $(R = 8.3\,J\,{K^{ - 1}}\,mo{l^{ - 1}})$View Solution
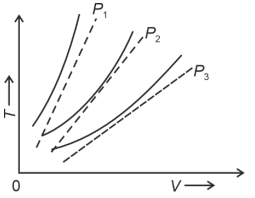
- 8The following graph represents the $T-V$ curves of an ideal gas (where $T$ is the temperature and $V$ the volume) at three pressures $P_1, P_2$ and $P_3$ compared with those of Charles's law represented as dotted lines.View Solution
Then the correct relation is:
- 9View SolutionA tyre kept outside in sunlight bursts off after sometime because of
- 10View SolutionVapour is injected at a uniform rate in a closed vessel which was initially evacuated. The pressure in the vessel
