A tyre kept outside in sunlight bursts off after sometime because of
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
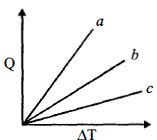
- 1Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
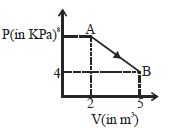
- 2A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
- 3The translational degrees of freedom $(f)$ and rotational degrees of freedom $(f)$ of $\mathrm{CH}_4$ molecule are:View Solution
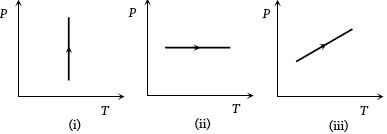
- 4View SolutionPressure versus temperature graphs of an ideal gas are as shown in figure. Choose the wrong statement
- 5The value of $C_V$ for one mole of neon gas isView Solution
- 6For a gas if $\gamma = 1.4$, then atomicity, ${C_p}$ and ${C_v}$ of the gas are respectivelyView Solution
- 7A thermally insulated rigid container of $1 \,L$ volume contains a diatomic ideal gas at room temperature. A small paddle installed inside the container is rotated from the outside, such that the pressure rises by $10^{5} \,Pa$. The change in internal energy is close to ............... $J$View Solution
- 8View SolutionAccording to the kinetic theory of gases, at absolute temperature
- 9An ideal gas with adiabatic exponent $(\gamma=1.5)$ undergoes a process in which work done by the gas is same as increase in internal energy of the gas. The molar heat capacity of gas for the process is -View Solution
- 10The temperature of an ideal gas is reduced from $927^\circ C$ to $27^\circ C$. The $r.m.s.$ velocity of the molecules becomesView Solution
