Assertion $(A):$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and volume.
Reason $(R):$ The molecules of gas collide with each other and the velocities of the molecules change due to the collision.
AIIMS 2015, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The change in volume $V$ with respect to an increase in pressure $P$ has been shown in the figure for a non-ideal gas at four different temperatures ${T_1},\,{T_2},\,{T_3}$ and ${T_4}$. The critical temperature of the gas isView Solution
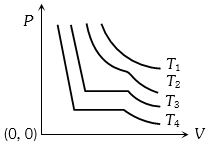
- 2View SolutionMoon has no atmosphere because
- 3View SolutionSaturated vapour is compressed to half its volume without any change in temperature, then the pressure will be
- 4$Assertion$: The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5\, times$ the product of its pressure and its volume.View Solution
$Reason$ : The molecules of a gas collide with each other and the velocities of the molecules change due to the collision. - 5The Mean Free Path $\ell$ for a gas molecule depends upon diameter, $d$ of the molecule asView Solution
- 6Two closed containers of equal volume filled with air at pressure $P_0$ and temperature $T_0$. Both are connected by a narrow tube. If one of the container is maintained at temperature $T_0$ and other at temperature $T$, then new pressure in the containers will beView Solution
- 7Equation of gas in terms of pressure $(P),$ absolute temperature $(T)$ and density $(d)$ isView Solution
- 8View SolutionAt a given volume and temperature, the pressure of a gas
- 9An air bubble doubles its radius on raising from the bottom of water reservoir to be the surface of water in it. If the atmospheric pressure is equal to $10\, m$ of water, the height of water in the reservoir is ..... $m$View Solution
- 10$40$ calories of heat is needed to raise the temperature of $1\, mole$ of an ideal monoatomic gas from $20°C$ to $30°C$ at a constant pressure. The amount of heat required to raise its temperature over the same interval at a constant volume $(R = 2\,calorie\,mol{e^{ - 1}}{K^{ - 1}})$ is ..... $calorie$View Solution
