$Assertion$: The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5\, times$ the product of its pressure and its volume.
$Reason$ : The molecules of a gas collide with each other and the velocities of the molecules change due to the collision.
$Reason$ : The molecules of a gas collide with each other and the velocities of the molecules change due to the collision.
AIIMS 2015, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a vessel, the gas is at pressure $P_0$. If the mass of all the molecules is halved and their speed is doubled, then the resultant pressure will beView Solution
- 2View SolutionThe degrees of freedom of a triatomic gas is
- 3When an air bubble of radius $‘r’$ rises from the bottom to the surface of a lake, its radius becomes $5r/4$ (the pressure of the atmosphere is equal to the $10 \,m$ height of water column). If the temperature is constant and the surface tension is neglected, the depth of the lake is .... $m$View Solution
- 4If pressure of $C{O_2}$ (real gas) in a container is given by $P = \frac{{RT}}{{2V - b}} - \frac{a}{{4{b^2}}}$ then mass of the gas in container is ...... $gm$View Solution
- 5At $NTP,$ sample of equal volume of chlorine and oxygen is taken. Now ratio of No. of moleculesView Solution
- 6A diatomic gas $(\gamma=1.4)$ does $100 \mathrm{~J}$ of work in an isobaric expansion. The heat given to the gas is :View Solution
- 7At a pressure of $24 \times {10^5}\,dyne/c{m^2}$, the volume of ${O_2}$ is $10\, litre$ and mass is $20\, gm.$ The $r.m.s.$ velocity will be....... $m/sec$View Solution
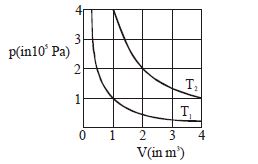
- 8The given diagram shows isotherms for a fixed mass of an ideal gas at temperature $T_1$ and $T_2$ . What is the value of the ratio $\frac{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_{2{\text{ }}}}}}{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_1}}}$ ?View Solution
- 9The molar specific heats of an ideal gas at constant pressure and volume are denoted by $C _{ P }$ and $C _{ V }$ respectively. If $\gamma=\frac{C_{p}}{C_{V}}$ and $R$ is the universal gas constant, then $C _{ V }$ is equal toView Solution
- 10The $r.m.s.$ velocity will be greater forView Solution
