$Assertion :$ Reversible systems are difficult to find in real world.
$Reason :$ Most processes are dissipative in nature.
$Reason :$ Most processes are dissipative in nature.
AIIMS 2005, Easy
In any process some energy is found to be converted into heat (dissipative in nature) due to which process becomes irreversible.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
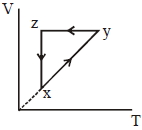
- 1A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
- 2A household refrigerator with a coefficient of performance $1.2$ removes heat from the refrigerated space at the rate of $60\ kJ/min$ .What would be cost of running this fridge for one month $\mathrm{Rs.}$ ..................... $(30\ days)$ (assuming each day it is used for $4$ hours and cost of one electrical unit is $6$ Rs.)View Solution
- 3View SolutionA perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
- 4An ideal gas at ${27^o}C$ is compressed adiabatically to $\frac{8}{{27}}$ of its original volume. If $\gamma = \frac{5}{3}$, then the rise in temperature is........ $K$View Solution
- 5In an isothermal reversible expansion, if the volume of $96\, gm$ of oxygen at $27°C$ is increased from $70$ litres to $140$ litres, then the work done by the gas will beView Solution
- 6In a certain thermodynamical process, the pressure of a gas depends on its volume as $kV ^{3}$. The work done when the temperature changes from $100^{\circ} C$ to $300^{\circ} C$ will be .......... $nR$, where $n$ denotes number of moles of a gas.View Solution
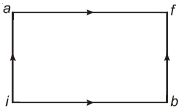
- 7When a system is taken from state $i$ to state $f$ along the path $iaf$, it is found that $Q=50$ $cal$ and $W=20$ $cal$ Along the path $ibf\ Q = 36\ cal. \ W$ along the path $ibf$ is ....... $ cal$View Solution
- 8Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
- 9View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
- 10A cyclic process $ABCA$ is shown in the $V-T$ diagram. Process on the $P-V$ diagram isView Solution