In an isothermal reversible expansion, if the volume of $96\, gm$ of oxygen at $27°C$ is increased from $70$ litres to $140$ litres, then the work done by the gas will be
Medium
(d) $W = \mu RT{\log _e}\frac{{{V_2}}}{{{V_1}}}$
$ = \left( {\frac{m}{M}} \right)RT{\log _e}\frac{{{V_2}}}{{{V_1}}} = 2.3 \times \frac{m}{M}RT{\log _{10}}\frac{{{V_2}}}{{{V_1}}}$
$ = 2.3 \times \frac{{96}}{{32}}R\,\,(273 + 27)\,{\log _{10}}\frac{{140}}{{70}} = \,2.3 \times 900R{\log _{10}}2$
$ = \left( {\frac{m}{M}} \right)RT{\log _e}\frac{{{V_2}}}{{{V_1}}} = 2.3 \times \frac{m}{M}RT{\log _{10}}\frac{{{V_2}}}{{{V_1}}}$
$ = 2.3 \times \frac{{96}}{{32}}R\,\,(273 + 27)\,{\log _{10}}\frac{{140}}{{70}} = \,2.3 \times 900R{\log _{10}}2$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 2View SolutionA reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
- 3At ${27^o}C$ a gas is suddenly compressed such that its pressure becomes $\frac{1}{8}th$ of original pressure. Temperature of the gas will be $(\gamma = 5/3)$View Solution
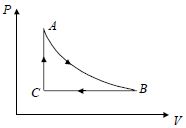
- 4A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
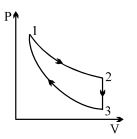
- 5Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
- 6A diatomic gas initially at $18^o C$ is compressed adiabatically to one-eighth of its original volume. The temperature after compression will beView Solution
- 7A monoatomic idea gas expands at constant pressure, with heat $Q$ supplied. The fraction of $Q$ which goes as work done by the gas isView Solution
- 8The adiabatic elasticity of a diatomic gas at $NTP$ is ........ $N / m ^2$View Solution
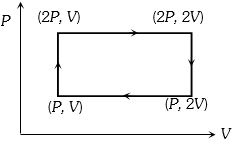
- 9Work done in the given $P-V$ diagram in the cyclic process isView Solution
- 10A Container having $1$ mole of a gas at a temperature $27°C$ has a movable piston which maintains at constant pressure in container of $1 \,atm.$ The gas is compressed until temperature becomes $127°C$. The work done is ........ $J$ ($C_P$ for gas is $7.03\, cal/mol-K)$View Solution
