A perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
Easy
(a) During free expansion of a perfect gas no, work is done and also no heat is supplied from outside. Therefore, no change in internal energy. Hence, temperature remain Hconstant.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If heat given to a system is $6 \,kcal$ and work done is $ 6 kJ.$ Then change in internal energy is ....... $kJ$View Solution
- 2View SolutionA gas is compressed adiabatically, which one of the following statement is NOT true.
- 3A Carnot freezer takes heat from water at $0\,^oC$ inside it and rejects it to the room at a temperature of $27\,^oC$. The latent heat of ice is $336 \times 10^3\, J\,kg^{-1}$. lf $5\, kg$ of water at $0\,^oC$ is converted into ice at $0\,^oC$ by the freezer, then the energy consumed by the freezer is close toView Solution
- 4$0.08 \mathrm{~kg}$ air is heated at constant volume through $5^{\circ} \mathrm{C}$. The specific heat of air at constant volume is $0.17 \mathrm{kcal} / \mathrm{kg}^{\circ} \mathrm{C}$ and $\mathrm{J}=4.18$ joule $/ \mathrm{cal}$. The change in its internal energy is approximately.View Solution
- 5A sample of $0.1\, g$ of water at $100^o C$ and normal pressure $(1.013 \times 10^5 N m^{-2} )$ requires $54\ cal $ of heat energy to convert to steam at $100^o C.$ If the volume of the steam produced is $167.1 \,cc,$ the change in internal energy of the sample, is ....... $J$View Solution
- 6For two different gases $X$ and $Y$, having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{V1}$ and $C_{V2}$ respectively, the ln $P$ versus ln $V$ graph is plotted for adiabatic process, as shownView Solution
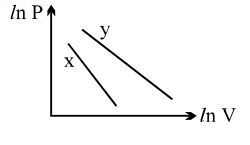
- 7A thermally insulated container is divided into two parts by a screen. In one part the pressure and temperature are $P$ and $T$ for an ideal gas filled. In the second part it is vacuum. If now a small hole is created in the screen, then the temperature of the gas willView Solution
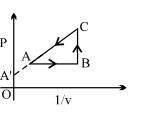
- 8View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
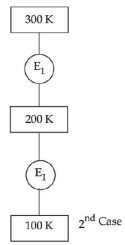
- 9In $1^{\text {st }}$ case, Carnot engine operates between temperatures $300\,K$ and $100\,K$. In $2^{\text {nd }}$ case, as shown in the figure, a combination of two engines is used. The efficiency of this combination (in $2^{\text {ad }}$ case) will be.View Solution
- 10$N$ moles of an ideal diatomic gas are in a cylinder at temperature $T$. suppose on supplying heat to the gas, its temperature remain constant but $n$ moles get dissociated into atoms. Heat supplied to the gas isView Solution
