The molar specific heats of an ideal gas at constant pressure and volume are denoted by $C _{ P }$ and $C _{ V }$ respectively. If $\gamma=\frac{C_{p}}{C_{V}}$ and $R$ is the universal gas constant, then $C _{ V }$ is equal to
AIPMT 2013, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
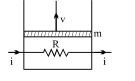
- 1A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution
- 2The ratio of mean kinetic energy of hydrogen and nitrogen at temperature $300 \,K$ and $450\, K$ respectively isView Solution
- 3The correct relation between $\gamma=\frac{ C _p}{ c _v}$ and temperature $T$ is :View Solution
- 4An ideal gas is enclosed in a container of volume $V$ at a pressure $P$. It is being pumped out of the container by using a pump with stroke volume $v$. What is final pressure in container after $n$-stroke of the pump? (assume temperature remains same)View Solution
- 5Two moles of ideal helium gas are in a rubber balloon at $30^o C$. The balloon is fully expandable and can be assumed to required no energy in its expansion. The temperature of the gas in the balloon is slowly changed to $35^o C$. The amount of heat required in raising the temperature is nearly...... $J$View Solution
(take $R$ $=$ $8.31$ $J/mol.K$) - 6Two vessels $A$ and $B$ are of the same size and are at same temperature. A contains $1 \mathrm{~g}$ of hydrogen and $B$ contains $1 \mathrm{~g}$ of oxygen. $\mathrm{P}_{\mathrm{A}}$ and $\mathrm{P}_{\mathrm{B}}$ are the pressures of the gases in $A$ and $\mathrm{B}$ respectively, then $\frac{\mathrm{P}_{\mathrm{A}}}{\mathrm{P}_{\mathrm{B}}}$ is:View Solution
- 7View SolutionA closed compartment containing gas is moving with some acceleration in horizontal direction. Neglect effect of gravity. Then the pressure in the compartment is
- 8View SolutionThe temperature below which a gas should be cooled, before it can be liquified by pressure only is termed as
- 9At what temperature will the oxygen molecules have the same root mean square speed as hydrogen molecules at $200 \,K$ ....... $K$View Solution
- 10Assertion $(A):$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and volume.View Solution
Reason $(R):$ The molecules of gas collide with each other and the velocities of the molecules change due to the collision.
