At a given temperature if ${V_{rms}}$ is the root mean square velocity of the molecules of a gas and ${V_s}$ the velocity of sound in it, then these are related as $\left( {\gamma = \frac{{{C_P}}}{{{C_v}}}} \right)$
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
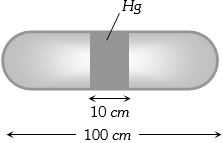
- 1A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 2A cylinder contains hydrogen gas at pressure of $249\; kPa$ and temperature $27^{\circ} C$. Its density is$.............kg / m ^{3}$ $\left( R =8.3\, J\, mol ^{-1} K ^{-1}\right)$View Solution
- 3The average distance between molecules of an ideal gas at $STP$ is approximately of the order ofView Solution
- 4The ratio of specific heats of a gas is $\frac{9}{7}$, then the number of degrees of freedom of the gas molecules for translational motion is :View Solution
- 5View SolutionIf two moles of diatomic gas and one mole of mono atomic gas are mixed then the ratio of specific heats is
- 6When water is heated from $0$ to $4\,^oC$View Solution
- 7Sound travels in a mixture of two moles of helium and n moles of hydrogen. If rms speed of gas molecules in the mixture is $\sqrt{2}$ times the speed of sound, then the value of $n$ will beView Solution
- 8A container contains $32 \,g$ of $O _2$ at a temperature $T$. The pressure of the gas is $P$. An identical container containing $4 g$ of $H _2$ at a temperature $2 T$ has a pressure of ............View Solution
- 9The temperature of an open room of volume $30\ m^3$ increases from $17^o C$ to $27vC$ due to sunshine. The atmospheric pressure in the room remains $1 \times 10^5\ Pa$. Ifni and nr are the number of molecules in the room before and after heating, then $n_f-n_i$ will be :View Solution
- 10View SolutionThe molecules of an ideal gas at a certain temperature have
