A cylinder contains hydrogen gas at pressure of $249\; kPa$ and temperature $27^{\circ} C$. Its density is$.............kg / m ^{3}$ $\left( R =8.3\, J\, mol ^{-1} K ^{-1}\right)$
NEET 2020, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At a given temperature the ratio of $r.m.s.$ velocities of hydrogen molecule and helium atom will beView Solution
- 2The average speed $v$ and $r.m.s.$ speed $\bar v$ of the molecules are related asView Solution
- 3The specific heat of $1$ mole of an ideal gas at constant pressure $({C_P})$ and at constant volume $({C_V})$ which is correctView Solution
- 4View SolutionFor matter to exist simultaneously in gas and liquid phases
- 5View SolutionThe expansion of unit mass of a perfect gas at constant pressure is shown in the diagram. Here
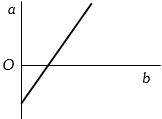
- 6What is/are the same for $O_2$ and $NH_3$ in gaseous stateView Solution
- 7Consider a gas with density $\rho $ and $\bar c$ as the root mean square velocity of its molecules contained in a volume. If the system moves as whole with velocity $v,$ then the pressure exerted by the gas isView Solution
- 8A gas is enclosed in a vessel at a constant temperature at a pressure of $5$ atmosphere and volume $4$ litre. Due to a leakage in the vessel, after some time, the pressure is reduced to $4$ atmosphere. As a result, theView Solution
- 9The respective speeds of the molecules are $1, 2, 3, 4$ and $5\, km/sec.$ The ratio of their $r.m.s. $ velocity and the average velocity will beView Solution
- 10In the kinetic theory of gases, which of these statements is/are true ?View Solution
$(i)$ The pressure of a gas is proportional to the mean speed of the molecules.
$(ii)$ The root mean square speed of the molecules is proportional to the pressure.
$(iii)$ The rate of diffusion is proportional to the mean speed of the molecules.
$(iv)$ The mean translational kinetic energy of a gas is proportional to its kelvin temperature.
