At what temperature $r.m.s.$ speed of air molecules doubles of that at $N.T.P.$ is ...... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Match the $\frac{C _{ P }}{ C _{ v }}$ ratio for ideal gases with different type of moleculesView Solution
Moleculae type $\frac{C _{ P }}{ C _{ v }}$ $(A)$ Monoatomic $(I)$ $\frac{7}{ 5}$ $(B)$ Diatomic rigid molecules $(II)$ $\frac{9}{7}$ $(C)$ Diatomic non-rigid molecules $(III)$ $\frac{4}{3}$ $(D)$ Triatomic rigid molecules $(IV)$ $\frac{5}{3}$ - 2View SolutionThe absolute zero is the temperature at which
- 3The kinetic energy of one gm-mole of a gas at normal temperature and pressure is $(R = 8.31 J/Mole-K)$View Solution
- 4The temperature at which the average translational kinetic energy of a molecule is equal to the energy gained by an electron in accelerating from rest through a potential difference of $1\, volt$ isView Solution
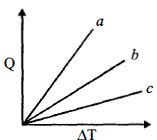
- 5Figure shows the variation in temperature $\left( {\Delta T} \right)$ with the amount of heat supplied $(Q)$ in an isobaric process corresponding to a monoatomic $(M)$, diatomic $(D)$ and a polyatomic $(P)$ gas. The initial state of all the gases are the same and the scales for the two axes coincide. Ignoring vibrational degrees of freedom, the lines $a, b$ and $c$ respectively correspond toView Solution
- 6The average kinetic energy of a helium atom at ${30^o}C$ isView Solution
- 7The molecules of a given mass of a gas have a $r.m.s.$ velocity of $200\, m/sec$ at $27°C$ and $1.0 \times {10^5}\,N/{m^2}$ pressure. When the temperature is $127°C$ and pressure is $0.5 \times {10^5}\,N/{m^2}$, the $r.m.s.$ velocity in $m/sec$ will beView Solution
- 8The equation for an ideal gas is $PV = RT,$ where $V$ represents the volume ofView Solution
- 9The $r.m.s.$ velocity will be greater forView Solution
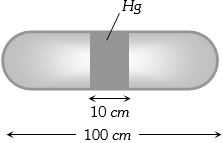
- 10A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution