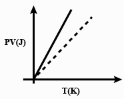
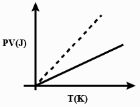
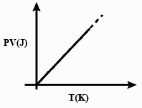
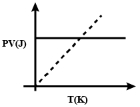
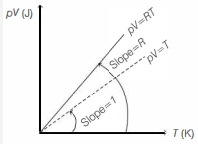
Which one of the following schematic graphs best represents the variation of $p V$ (in Joules) versus $T$ (in Kelvin) of one mole of an ideal gas? (The dotted line represents $p V=T$ )
KVPY 2019, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe time average of the kinetic energy of one molecule of a gas taken over a long period of time
- 2View SolutionThe number of translational degrees of freedom for a diatomic gas is
- 3View SolutionThe temperature below which a gas should be cooled, before it can be liquified by pressure only is termed as
- 4At a temperature $T K,$ the pressure of $4.0\,\,g$ argon in a bulb is $p$. The bulb is put in a bath having temperature higher by $50\,\,K$ than the first one. $0.8\,\,g$ of argon gas had to be removed to maintained original pressure. The temperature $T$ is equal to ..... $K$View Solution
- 5A vessel contains $14\,g$ of nitrogen gas at a temperature of $27^{\circ}\,C$. The amount of heat to be transferred to the gap to double the r.m.s. speed of its molecules will be $......J$ $\left(\right.$ Take $R =8.32\,J\,mol ^{-1} k ^{-1}$ )View Solution
- 6The number of gas molecules striking per second per square metre of the top surface of a table placed in a room at $20^{\circ} C$ and 1 atmospheric pressure is of the order of $\left(k_{B}=1.4 \times 10^{-23} \,JK ^{-1}\right.$ and the average mass of an air molecule is $5 \times 10^{-27} \,kg$ )View Solution
- 7Two ideal gases at absolute temperature $T_1$ and $T_2$ are mixed. There is no loss of energy. The masses of the molecules are $m_1$ and $m_2$ and the number of molecules in the gases are $n_1$ and $n_2$ respectively. The temperature of mixture will beView Solution
- 8The air density at Mount Everest is less than that at the sea level. It is found by mountaineers that for one trip lasting a few hours, the extra oxygen needed by them corresponds to $30,000\ cc$ at sea level (pressure $1$ atmosphere, temperature $27^oC).$ Assuming that the temperature around Mount Everest is $-73^oC$ and that the oxygen cylinder has capacity of $5.2\ litre,$ the pressure at which $O_2$ be filled (at site) in cylinder is ..... $atm$View Solution
- 9$2\, moles$ of helium gas is mixed with $3\, moles$ of hydrogen molecules (taken to be rigid). What is the molar specific heat of mixture at constant volume ....... $J/mol\, K$ ? $(R = 8.3\, J/mol\, K)$View Solution
- 10The specific heats, $C_P$ and $C_V$ of a gas of diatomic molecules, $A$, are given (in units of $J\, mol^{-1}\, K^{-1}$) by $29$ and $22$, respectively. Another gas of diatomic molecules $B$, has the corresponding values $30$ and $21$. If they are treated as ideal gases, thenView Solution