Butane has a _______ boiling point to that of propanol.
- Lower
The molecules of butane are held together by weak vander Waals forces of attraction, while those of propanol are held together by stronger intermolecular hydrogen bonding.
Therefore, the boiling point of propanol is much higher than of butane.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhich of the following alcohols is the least soluble in water?
- 2View SolutionWhich of the following name does not fit a real name?
- 3View SolutionTrivial name of methanol is _________.
- 4View SolutionDehydration of the following in increasing order is:
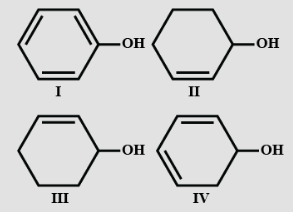
- 5View SolutionWhat is the hybridisation of carbon and oxygen in electronic structure of ether?
- 6View SolutionWhich of the following can not be the product of this reaction?
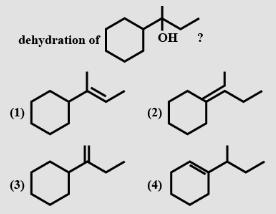
- 7View Solution
- 8$\ce{CH_3CH_2OH}$ can be converted into $\ce{CH_3CHO}$ by $.........$View Solution
- 9View SolutionThe major product obtained on acid - catalysed hydration of 2-phenylpropene is:
- 10Phenol reacts with bromine water in carbon disulphate at low temperature to give$:$View Solution

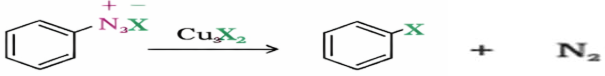 is-
is-