The temperature inside and outside of refrigerator are $260\, K$ and $315\, K$ respectively. Assuming that the refrigerator cycle is reversible, calculate the heat delivered to surroundings for every joule of work done.
Medium
$\mathrm{T}_{2}=260 \mathrm{K} . \mathrm{T}_{1}=315 \mathrm{K} \quad ; \mathrm{W}=1$ joule
Coefficient of performance of Carnot
refrigerator $\beta=\frac{\mathrm{Q}_{2}}{\mathrm{W}}=\frac{\mathrm{T}_{2}}{\mathrm{T}_{1}-\mathrm{T}_{2}}$
$\therefore \frac{\mathrm{Q}_{2}}{1}=\frac{260}{315-260}=\frac{260}{55} \Rightarrow$
$\mathrm{Q}_{2}=\frac{260}{55}=4.73 \mathrm{J}$
$\mathrm{Q}_{1}=\mathrm{Q}_{2}+\mathrm{W}=5.73$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic gas $(\gamma=1.4)$ does $400 J$ of work when it is expanded isobarically. The heat given to the gas in the process is ............ $J$View Solution
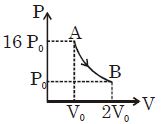
- 2Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
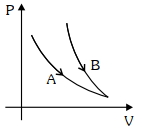
- 3Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
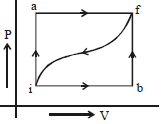
- 4When a system is taken from a state $i$ to $f$ along the path $iaf$ (as shown in the figure). $Q = 50\, cal$ and $W = 20\, cal$ ; along path $ibf,$ $Q = 36\, cal.$View Solution
$(i)$ What is $W$ along path $ibf$ ?
$(ii)$ If $W = 13$ cal for path $fi$, what is $Q$ for the path $fi$ ?
$(iii)$ Take $E_{int,i} = 10\,\, cal$ then what is $E_{int,f}$ ?
- 5The latent heat of vaporization of water is $2240 \,J/gm$. If the work done in the process of vaporization of $1\, gm$ is $168\, J$, then increase in internal energy is .... $J$View Solution
- 6Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
- 7View SolutionThe ratio of work done by an ideal monoatomic gas to the heat supplied to it in an isobaric process is
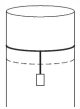
- 8A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 9View SolutionSelect the correct statement for work, heat and change in internal energy.
- 10Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ isView Solution