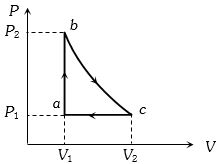
Carbon monoxide is carried around a closed cycle $abc$ in which bc is an isothermal process as shown in the figure. The gas absorbs $7000 J$ of heat as its temperature increases from $300 K$ to $1000 K$ in going from $a$ to $b$. The quantity of heat rejected by the gas during the process $ca$ is ..... $J$

Diffcult
(d) For path $ab : {(\Delta U)_{ab}} = 7000\;J$
By using $\Delta U = \mu {C_V}\Delta T$
$7000 = \mu \times \frac{5}{2}R \times 700 \Rightarrow \mu = 0.48$
For path $ca : {(\Delta Q)_{ca}} = {(\Delta U)_{ca}} + {(\Delta W)_{ca}}$ ….$(i)$
${(\Delta U)_{ab}} + {(\Delta U)_{bc}} + {(\Delta U)_{ca}} = 0$
$\therefore $$7000 + 0 + {(\Delta U)_{ca}} = 0 \Rightarrow {(\Delta U)_{ca}} = - 7000\;J$ ...$(ii)$
Also ${(\Delta W)_{ca}} = {P_1}({V_1} - {V_2}) = \mu R({T_1} - {T_2})$
$ = 0.48 \times 8.31 \times (300 - 1000) = - 2792.16\;J$ ….$(iii)$
on solving equations $(i), (ii)$ and $ (iii)$
${(\Delta Q)_{ca}} = - 7000 - 2792.16 = - 9792.16\;J$$ = - 9800\;J$
By using $\Delta U = \mu {C_V}\Delta T$
$7000 = \mu \times \frac{5}{2}R \times 700 \Rightarrow \mu = 0.48$
For path $ca : {(\Delta Q)_{ca}} = {(\Delta U)_{ca}} + {(\Delta W)_{ca}}$ ….$(i)$
${(\Delta U)_{ab}} + {(\Delta U)_{bc}} + {(\Delta U)_{ca}} = 0$
$\therefore $$7000 + 0 + {(\Delta U)_{ca}} = 0 \Rightarrow {(\Delta U)_{ca}} = - 7000\;J$ ...$(ii)$
Also ${(\Delta W)_{ca}} = {P_1}({V_1} - {V_2}) = \mu R({T_1} - {T_2})$
$ = 0.48 \times 8.31 \times (300 - 1000) = - 2792.16\;J$ ….$(iii)$
on solving equations $(i), (ii)$ and $ (iii)$
${(\Delta Q)_{ca}} = - 7000 - 2792.16 = - 9792.16\;J$$ = - 9800\;J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1One mole of a gas obeying the equation of state $P(V-b)=R T$ is made to expand from a state with coordinates $\left(P_{1}, V_{1}\right)$ to a state with $\left(P_{2}, V_{2}\right)$ along a process that is depicted by a straight line on a $P-V$ diagram. Then, the work done is given byView Solution
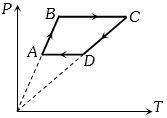
- 2Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$View Solution
- 3View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
- 4A monatornic gas at a pressure $P,$ having a volume $V$ expands isothermally to a volume $2\, V$ and then adiabatically to a volume $16\, V.$ The final pressure of the gas is $(\,Take \,\gamma = 5/3)$View Solution
- 5The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.View Solution
- 6View SolutionWhen a gas expands adiabatically
- 7The efficiency of an ideal heat engine working between the freezing point and boiling point of water, is ........ $\%$View Solution
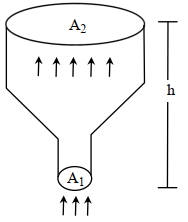
- 8An ideal gas of density $\rho=0.2 kg m ^{-3}$ enters a chimney of height $h$ at the rate of $\alpha=0.8 kg s ^{-1}$ from its lower end, and escapes through the upper end as shown in the figure. The cross-sectional area of the lower end is $A_1=0.1 m ^2$ and the upper end is $A_2=0.4 m ^2$. The pressure and the temperature of the gas at the lower end are $600 Pa$ and $300 K$, respectively, while its temperature at the upper end is $150 K$. The chimney is heat insulated so that the gas undergoes adiabatic expansion. Take $g=10 ms ^{-2}$ and the ratio of specific heats of the gas $\gamma=2$. Ignore atmospheric pressure.View Solution
Which of the following statement($s$) is(are) correct?
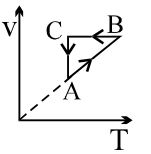
- 9An ideal gas undergoes cyclic process as shown in density pressure graph. During the process $AB$ the work done $|W_{AB}| = 70\,J$ . During the process $BC$, the gas absorbs $150\,J$ of heat. During the process $CA$ , gas undergoes expansion and does $210\,J$ of workView Solution
- 10The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$View Solution
