The efficiency of an ideal heat engine working between the freezing point and boiling point of water, is ........ $\%$
NEET 2018,JEE MAIN 2022, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
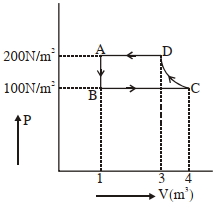
- 1The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)View Solution
- 2A polyatomic gas with six degrees of freedom does $25\,\,J$ of work when it is expanded at constant pressure. The heat given to the gas is ..... $J$View Solution
- 3A diatomic ideal gas is used in a Carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\ V$, the efficiency of the engine isView Solution
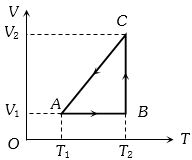
- 4A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
- 5In a thermodynamic process pressure of a fixed mass of a gas is changed in such a manner that the gas releases $30$ joules of heat and $10$ joules of work was done on the gas. If the initial internal energy of the gas was $30$ joules, then the final internal energy will be ........ $J$View Solution
- 6A monoatomic ideal gas, initially at temperature ${T_1},$ is enclosed in a cylinder fitted with a frictionless piston. The gas is allowed to expand adiabatically to a temperature. ${T_2}$ by releasing the piston suddenly. If ${L_1}$ and ${L_2}$ are the lengths of the gas column before and after expansion respectively, then ${T_1}/{T_2}$ is given byView Solution
- 7View SolutionThe slopes of isothermal and adiabatic curves are related as
- 8View SolutionFor adiabatic process, wrong statement is
- 9A diatomic ideal gas is used in a carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\,V$ , the efficiency of the engine isView Solution
- 10Following figure shows on adiabatic cylindrical container of volume ${V_0}$ divided by an adiabatic smooth piston (area of cross-section = $A$ ) in two equal parts. An ideal gas $({C_P}/{C_V} = \gamma )$ is at pressure $P_1$ and temperature $T_1$ in left part and gas at pressure $P_2$ and temperature $T_2$ in right part. The piston is slowly displaced and released at a position where it can stay in equilibrium. The final pressure of the two parts will be (Suppose $ x$ = displacement of the piston)View Solution