Consider one mole of helium gas enclosed in a container at initial pressure $P_1$ and volume $V_1$. It expands isothermally to volume $4 V_1$. After this, the gas expands adiabatically and its volume becomes $32 V_1$. The work done by the gas during isothermal and adiabatic expansion processes are $W_{\text {iso }}$ and $W_{\text {adia, }}$ respectively. If the ratio $\frac{W_{\text {iso }}}{W_{\text {adia }}}=f \ln 2$, then $f$ is. . . . . . . .
IIT 2020, Medium
$\frac{ P _1}{4}\left(4 V _1\right)^{5 / 3}= P _2\left(32 v _1\right)^{5 / 3}$
$P _2=\frac{ P _1}{4}\left(\frac{1}{8}\right)^{5 / 3}=\frac{ P _1}{128}$
$W _{ adi }=\frac{ P _1 V _1- P _2 V _2}{\gamma-1}=\frac{ P _1 V _1-\frac{P_1}{128}\left(32 V _1\right)}{\frac{5}{3}-1}$
$=\frac{P_1 V_1(3 / 4)}{2 / 3}=\frac{9}{8} P_1 V_1$
$W _{ iso } \quad= P _1 V _1 \operatorname{In}\left(\frac{4 V _1}{ V _1}\right)=2 P _1 V _1 \ln 2$
$\frac{ W _{ iso }}{ W _{\text {zdio }}}=\frac{2 P _1 V _1 \ln 2}{\frac{9}{8} P _1 V _1}=\frac{16}{9} \ln 2= f \ln 2$
$f =\frac{16}{9}=1.7778 \approx 1.78$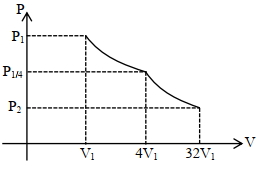
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
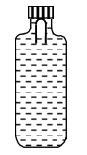
- 1A soft plastic bottle, filled with water of density $1 gm / cc$, carries an inverted glass test-tube with some air (ideal gas) trapped as shown in the figure. The test-tube has a mass of $5 gm$, and it is made of a thick glass of density $2.5 gm / cc$. Initially the bottle is sealed at atmospheric pressure $p_0=10^5 Pa$ so that the volume of the trapped air is $v_0=3.3 cc$. When the bottle is squeezed from outside at constant temperature, the pressure inside rises and the volume of the trapped air reduces. It is found that the test tube begins to sink at pressure $P_0+\Delta p$ without changing its orientation. At this pressure, the volume of the trapped air is $v_0-\Delta v$.View Solution
Let $\Delta v=X$ cc and $\Delta p=Y \times 10^3 Pa$.
($1$) The value of $X$ is
($2$) The value of $Y$ is
Give the answer or quetion ($1$) and ($2$)
- 2A perfect gas goes from state $A$ to another state $B$ by absorbing $8 \times {10^5}J$ of heat and doing $6.5 \times {10^5}J$ of external work. It is now transferred between the same two states in another process in which it absorbs ${10^5}J$ of heat. Then in the second processView Solution
- 3An ideal gas is made to undergo the cyclic process shown in the figure below. Let $\Delta W$ depict the work done, $\Delta U$ be the change in internal energy of the gas and $Q$ be the heat added to the gas. Sign of each of these three quantities for the whole cycle will be (0 refers to no change)View Solution
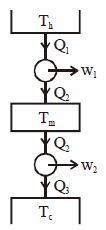
- 4Suppose that two heat engines are connected in series, such that the heat exhaust of the first engine is used as the heat input of the second engine as shown in figure. The efficiencies of the engines are $\eta_1$ and $\eta_2$, respectively. The net efficiency of the combination is given byView Solution
- 5The pressure and volume of a gas are changed as shown in the $P-V$ diagram in this figure. The temperature of the gas will ........View Solution
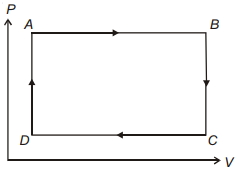
- 6The three processes in a thermodynamic cycle shown in the figure are : Process $1 \rightarrow 2$ is isothermal; Process $2 \rightarrow 3$ is isochoric (volume remains constant); Process $3 \rightarrow 1$ is adiabatic. The total work done by the ideal gas in this cycle is $10 \,J$. The internal energy decreases by $20 \,J$ in the isochoric process. The work done by the gas in the adiabatic process is $-20 \,J$. The heat added to the system in the isothermal process is .............. $J$View Solution
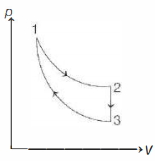
- 7View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
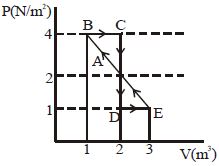
- 8One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
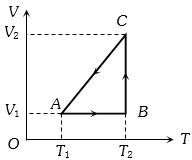
- 9A cyclic process for $1\, mole$ of an ideal gas is shown in figure in the $V-T,$ diagram. The work done in $AB, BC$ and $CA$ respectivelyView Solution
- 10An ideal gas follows a process $PT =$ constant. The correct graph between pressure $\&$ volume isView Solution
