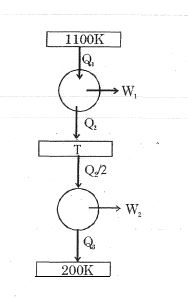
Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)

Diffcult
$\mathrm{Q}_{1}-\mathrm{Q}_{2}=\frac{\mathrm{Q}_{2}}{2}-\mathrm{Q}_{3}$
$\frac{Q_{1}}{Q_{2}}-1=\frac{1}{2}-\frac{Q_{3}}{Q_{2}}=\eta-1=\frac{1}{2}-\frac{\eta^{\prime}}{2}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionFor free expansion of the gas which of the following is true
- 2Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$View Solution
- 3View SolutionEntropy of a system decreases .........
- 4In an adiabatic process, the density of a diatomic gas becomes $32$ times its initial value. The final pressure of the gas is found to be $n$ times the initial pressure. The value of $n$ isView Solution
- 5A cylinder fitted with a piston contains $0.2 \,moles$ of air at temperature $27°C.$ The piston is pushed so slowly that the air within the cylinder remains in thermal equilibrium with the surroundings. Find the approximate work done by the system if the final volume is twice the initial volume ...... $J$View Solution
- 6View SolutionSelect the correct statement for work, heat and change in internal energy.
- 7$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
$Reason :$ Entropy increases in all natural processes. - 8View SolutionThe maximum possible efficiency of a heat engine is ...........
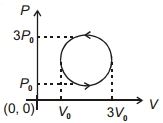
- 9View SolutionWork done in the cyclic process shown in figure is ...........
- 10An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
