The maximum possible efficiency of a heat engine is ...........
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
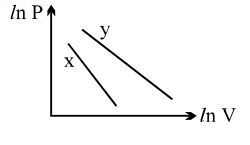
- 1For two different gases $X$ and $Y$, having degrees of freedom $f_1$ and $f_2$ and molar heat capacities at constant volume $C_{V1}$ and $C_{V2}$ respectively, the ln $P$ versus ln $V$ graph is plotted for adiabatic process, as shownView Solution
- 2A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 3In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
- 4A monoatomic gas at pressure $P$ and volume $V$ is suddenly compressed to one eighth of its original volume. The final pressure at constant entropy will be $.....P$View Solution
- 5$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
$Reason :$ Dissipative effects cannot be eliminated. - 6View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
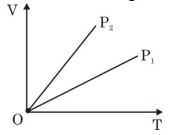
- 7View SolutionThe given figure represents two isobaric processes for the same mass of an ideal gas, then
- 8Work done by air when it expands from $50\, litres$ to $150\, litres$ at a constant pressure of $2$ atmosphere isView Solution
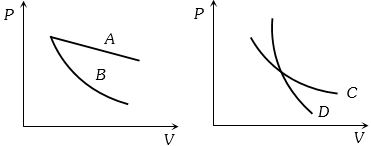
- 9In the following figure, four curves $A, B, C$ and $D$ are shown. The curves areView Solution
- 10Find the amount of work done to increase the temperature of one mole of an ideal gas by $30^o\ C$ if it is expanding under the condition .... $J$View Solution
$V\propto {T^{\frac{2}{3}}}$ $[R = 1.99\ cal/mol-K]$
