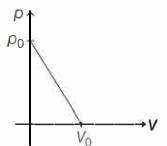
One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ is

KVPY 2015, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If the ratio of specific heat of a gas at constant pressure to that at constant volume is $\gamma $, the change in internal energy of a mass of gas, when the volume changes from $V$ to $2V$ constant pressure $ p$, isView Solution
- 2A thermodynamic process in which temperature $T$ of the system remains constant though other variable $P$ and $V$ may change, is calledView Solution
- 3View SolutionWhich of the following is not thermodynamical function
- 4One mole of a monoatomic ideal gas $\left(c_{ V }=\frac{3}{2} R \right)$ undergoes a cycle where it first goes isochorically from the state $\left(\frac{3}{2} P _0, V _0\right)$ to $\left( P _0, V _0\right)$, and then is isobarically contracted to the volume $\frac{1}{2} V _0$. It is then taken back to the initial state by a path which is a quarter ellipse on the $P - V$ diagram. The efficiency of this cycle isView Solution
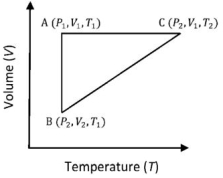
- 5A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
- 6The change in the entropy of a $1$ mole of an ideal gas which went through an isothermal process from an initial state $(P_1, V_1,T)$ to the final state $(P_2, V_2,T)$ is equal toView Solution
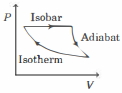
- 7The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
- 8The $P-V$ diagram of a diatomic ideal gas system going under cyclic process as shown in figure. The work done during an adiabatic process $CD$ is (use $\gamma=1.4$) (in $J$)View Solution
- 9A certain mass of gas at $273 K$ is expanded to $81$ times its volume under adiabatic condition. If $\gamma = 1.25$ for the gas, then its final temperature is ..... $^oC$View Solution
- 10A Carnot engine whose sink is at $300 \,K$ has an efficiency of $50 \%$. By how much should the temperature of source be increased so as the efficiency becomes $70 \%$ is ............ $K$View Solution