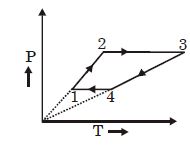
The work done during the isobaric processes $2-3$ and $1-4$ are as follows:
$\mathrm{W}_{2-3}=\mathrm{P}_{2}\left(\mathrm{V}_{3}-\mathrm{V}_{2}\right)$
$w_{1-4}=P_{1}\left(V_{1}-v_{4}\right)$
Total work done $=\mathrm{P}_{2}\left(\mathrm{V}_{3}-\mathrm{V}_{2}\right)+\mathrm{P}_{1}\left(\mathrm{V}_{1}-\mathrm{V}_{4}\right)$
$\mathrm{W}_{\mathrm{T}}=\mathrm{P}_{2} \mathrm{V}_{3}-\mathrm{P}_{2} \mathrm{V}_{2}+\mathrm{P}_{1} \mathrm{V}_{1}-\mathrm{P}_{1} \mathrm{V}_{4}$
Three moles has been given, so
$\mathbf{P V}=n R T=3 R T$
$\mathrm{W}_{\mathrm{T}}=3 \mathrm{RT}_{3}-3 \mathrm{RT}_{2}+3 \mathrm{RT}_{1}-3 \mathrm{RT}_{4}$
$=3 \mathrm{R}\left[\mathrm{T}_{1}+\mathrm{T}_{3}-\mathrm{T}_{2}-\mathrm{T}_{4}\right]$
$=3 \mathrm{R}[400+2400-800-1200]$
$=3 \mathrm{R} \times 800=20 \times 10^{3} \mathrm{J}=20 \mathrm{kJ}$
Download our appand get started for free
Similar Questions
- 1Match List $I$ with List $II$ :View Solution
List $I$ List $II$ $A$ Isothermal Process $I$ Work done by the gas decreases internal energy $B$ Adiabatic Process $II$ No change in internal energy $C$ Isochoric Process $III$ The heat absorbed goes partly to increase internal energy and partly to do work $D$ Isobaric Process $IV$ No work is done on or by the gas Choose the correct answer from the options given below :
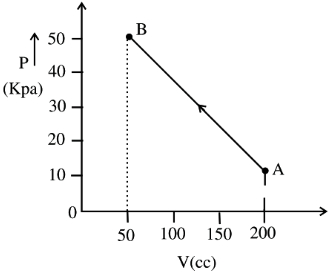
- 2The pressure of a gas changes linearly with volume from $A$ to $B$ as shown in figure. If no heat is supplied to or extracted from the gas then change in the internal energy of the gas will be $............\,J$View Solution
- 3View SolutionWhen a gas expands adiabatically
- 4View SolutionWhich of the following statements is correct for any thermodynamic system
- 5An ideal gas at ${27^o}C$ is compressed adiabatically to $\frac{8}{{27}}$ of its original volume. If $\gamma = \frac{5}{3}$, then the rise in temperature is........ $K$View Solution
- 6If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
- 7For an adiabatic expansion of an ideal gas, the fractional change in its pressure is equal to (where $\gamma$ is the ratio of specific heats):View Solution
- 8View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
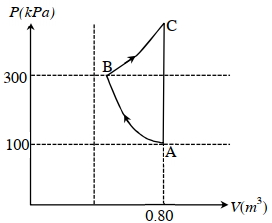
- 9In the given $P$-V diagram, a monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is first compressed adiabatically from state $A$ to state $B$. Then it expands isothermally from state $B$ to state $C$. [Given: $\left(\frac{1}{3}\right)^{0.6} \simeq 0.5, \ln 2 \simeq 0.7$ ].View Solution
Which of the following statement($s$) is(are) correct?
$(A)$ The magnitude of the total work done in the process $A \rightarrow B \rightarrow C$ is $144 kJ$.
$(B)$ The magnitude of the work done in the process $B \rightarrow C$ is $84 kJ$.
$(C)$ The magnitude of the work done in the process $A \rightarrow B$ is $60 kJ$.
$(D)$ The magnitude of the work done in the process $C \rightarrow A$ is zero.
- 10Diatomic gas is used in carnot heat engine. If efficiency of given carnot heat engine is $80\%$ , then find the ratio of initial volume to final volume of gas during adiabatic expansionView Solution
