A diatomic ideal gas is used in a Carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\ V$, the efficiency of the engine is
AIEEE 2010, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
- 2Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

- 3A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) isView Solution
- 4The figure given below shows the variation in the internal energy $U$ with volume $V$ of $2.0\ mole$ of an ideal gas in a cyclic process $abcda$ . The temperatures of the gas during the processes $ab$ and $cd$ are $500\ K$ and $300\ K$ respectively, the heat absorbed by the gas during the complete process is .... $J$View Solution
(Take $R$ = $8.3\ J/mol-K$ and $ln\ 2$ = $0.69$)
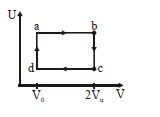
- 5One mole of an ideal gas in initial state $\mathrm{A}$ undergoes a cyclic process $A B C A$, as shown in the figure. Its pressure at $A$ is $\mathrm{P}_0$. Choose the correct option$(s)$ from the followingView Solution
$(A)$ Internal energies at $\mathrm{A}$ and $\mathrm{B}$ are the same
$(B)$ Work done by the gas in process $\mathrm{AB}$ is $\mathrm{P}_0 \mathrm{~V}_0 \ln 4$
$(C)$ Pressure at $C$ is $\frac{P_0}{4}$
$(D)$ Temperature at $\mathrm{C}$ is $\frac{\mathrm{T}_0}{4}$
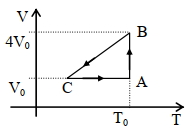
- 6A sample of gas expands from volume ${V_1}$ to ${V_2}$. The amount of work done by the gas is greatest when the expansion isView Solution
- 7Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
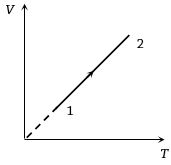
- 8View SolutionIn thermodynamics, heat and work are
- 9A Carnot engine has efficiency of $50 \%$. If the temperature of sink is reduced by $40^{\circ} C$, its efficiency increases by $30 \%$. The temperature of the source will be$....K$View Solution
- 10View SolutionWhich of the following statements is correct for any thermodynamic system
