During dehydration of alcohols to alkenes by heating with conc. $\ce{H_2SO_{4}},$ the initiation step is$:$
Dehydration of alcohol to alkene in presence of concentrated $\ce{H_2SO_{4}}$ involves following steps$:$
Thus, the initiation step is protonation of alcohol.
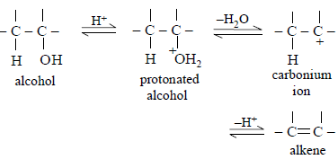
Thus, the initiation step is protonation of alcohol.

Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Reagents used for industrial preparation of methanol are$:$View Solution
- 2View SolutionAn ester can be prepared by the reaction of:
- 3View SolutionMethanol is called as Marine Methanol beacuse:
- 4When $3-$methylbutan$-2-$ol is treated with $\ce{HBr},$ the following reaction takes place. What are the correct steps for formation of the above product?View Solution
- 5What is the correct order of reactivity of alcohols in the following reaction?View Solution
$\text{R}-\text{OH}+\text{HCL}\xrightarrow{\text{ZnCl}_2}\text{R}-\text{Cl}+\text{H}_2\text{O}$ - 6View SolutionWhich of the following can not be the product of this reaction?
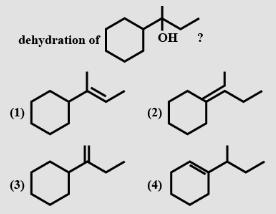
- 7View SolutionWhich of the following reactions is possible?
- 8Phenol reacts with bromine water in carbon disulphate at low temperature to give$:$View Solution
- 9Among the four compounds $(i)$ acetone, $(ii)$ propanol, $(iiii)$ methyl acetate and $(iv)$ propionic acid, the two that are isomeric are:View Solution
- 10When ethyl alcohol reacts with nitric acid, it forms$:$View Solution
