Figure shows a polytropic process for an ideal gas. The work done by the gas will be in process $AB$ is
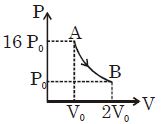

Medium
$\because P_{1} V_{1}^{x}=P_{2} V_{2}^{x}$
$\frac{16 \mathrm{P}_{0}}{\mathrm{P}_{0}}=\left(\frac{2 \mathrm{V}_{0}}{\mathrm{V}_{0}}\right)^{\mathrm{x}}$
$x=4$
$\mathrm{w}=\frac{\mathrm{P}_{2} \mathrm{V}_{2}-\mathrm{P}_{1} \mathrm{V}_{1}}{1-\mathrm{x}}=\frac{2 \mathrm{P}_{0} \mathrm{V}_{0}-16 \mathrm{P}_{0} \mathrm{V}_{0}}{1-4}$
$=\frac{14}{3} \mathrm{P}_{0} \mathrm{V}_{0}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
- 2A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
- 3View SolutionWhich of the following can not determine the state of a thermodynamic system
- 4View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure
- 5A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
- 6Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
- 7In thermodynamic process, $200$ Joules of heat is given to a gas and $100$ Joules of work is also done on it. The change in internal energy of the gas is ........ $J$View Solution
- 8Work done by a system under isothermal change from a volume ${V_1}$ to ${V_2}$ for a gas which obeys Vander Waal's equation $(V - \beta n)\,\left( {P + \frac{{\alpha {n^2}}}{V}} \right) = nRT$View Solution
- 9View SolutionIn a reversible isochoric change
- 10The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
