Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ If the internal energy of He sample of $100\,\,J$ and that of the hydrogen sample is $200\,\,J$, then the internal energy of the mixture is ..... $J$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two gases-argon (atomic radius $0.07 \;\mathrm{nm}$,atomic weight $40$ ) and xenon (atomic radius $0.1\; \mathrm{nm},$ atomic weight $140$ ) have the same number density and are at the same temperature. The raito of their respective mean free times is closest toView Solution
- 2The pressure of an ideal gas is written as $E = \frac{3PV}{2}$ . Here $E$ stands forView Solution
- 3A vessel contains $1$ mole of $O_2$ gas (molar mass $32$) at a temperature $T$. The pressure of the gas is $P$. An identical vessel containing one mole of $He$ gas (molar mass $4$) at a temperature $2T$ has a pressure ofView Solution
- 4$c_P$ and $c_V$ are specific heats at constant pressure and constant volume respectively. It is observed thatView Solution
$c_P - c_V = a$ for hydrogen gas
$c_P - c_V = b$ for nitrogen gas
The correct relation between $a$ and $b$ is
- 5From the following $V-T$ diagram we can concludeView Solution
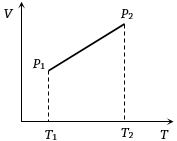
- 6At what temperature the molecules of nitrogen will have the same $r.m.s.$ velocity as the molecules of oxygen at $127°C$ ....... $^oC$View Solution
- 7A polyatomic ideal gas has $24$ vibrational modes. What is the value of $\gamma$ ?View Solution
- 8The average kinetic energy of a gas molecule at $27^oC$ is $6.21 \times {10^{ - 21}}\,J$. Its average kinetic energy at $227°C$ will beView Solution
- 9One mole of a gas mixture is heated under constant pressure, and heat supplied $Q$ is plotted against temperature difference acquired. Find the approximate value of $\gamma $ for mixtureView Solution
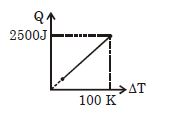
- 10Under constant temperature, graph between $P$ and $1/V$ isView Solution
