$c_P$ and $c_V$ are specific heats at constant pressure and constant volume respectively. It is observed that
$c_P - c_V = a$ for hydrogen gas
$c_P - c_V = b$ for nitrogen gas
The correct relation between $a$ and $b$ is
JEE MAIN 2017, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The equation of state corresponding to $8 \,g$ of ${O_2}$ isView Solution
- 2At which temperature the r.m.s. velocity of a hydrogen molecule equal to that of an oxygen molecule at $47^{\circ} \mathrm{C}$ ?View Solution
- 3An engineer is given a fixed volume $V_m$ of metal with which to construct a spherical pressure vessel. Interestingly, assuming the vessel has thin walls and is always pressurized to near its bursting point, the amount of gas the vessel can contain, $n$ (measured in moles), does not depend on the radius $r$ of the vessel : instead it depends only on $V_m$ (measured in $m^3$) the temperature $T$ (mensured in $K$). the ideal gas constant $R$ (measured in $J/(K\ mol$ )), and the tensile strength of the metal $\sigma $ (measured in $N/m^2$ ) . Which of the following gives $n$ in terms of these parameters?View Solution
- 4The mean kinetic energy of a gas at $300\, K$ is $100\, J.$ The mean energy of the gas at $450\, K$ is equal to ...... $J$View Solution
- 5A gas mixture consists of $2$ moles of $O_2$ and $4$ moles of $Ar$ at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
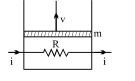
- 6A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution
- 7A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$View Solution
- 8Consider a mixture of gas molecule of types $A, B$ and $C$ having masses $m_{A}\,<\,m_{B}\,<\,m_{C}$ ratio of their root mean square speeds at normal temperature and pressure is :View Solution
- 9The average kinetic energy of a gas at $-23°C$ and $75\, cm$ pressure is $5 \times {10^{ - 14}}\,erg$ for ${H_2}$. The mean kinetic energy of the ${O_2}$ at $227°C$ and $150\, cm$ pressure will beView Solution
- 10A gas is collected over the water at $25°C.$ The total pressure of moist gas was $735 \,mm$ of mercury. If the aqueous vapour pressure at $25°C$ is $23.8\, mm.$ Then the pressure of dry gas is..... $mm$View Solution
