A closed container is fully insulated from outside. One half of it is filled with an ideal gas $X$ separated by a plate Pfrom the other half $Y$ which contains a vacuum as shown in figure. When $P$ is removed, $X$ moves into $Y$. Which of the following statements is correct?
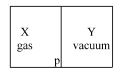

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
- 2Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be comeView Solution
- 3A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
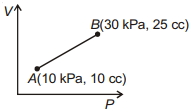
- 4Work done for the process shown in the figure is ............ $J$View Solution
- 5A gas expands adiabatically at constant pressure such that its temperature $T \propto \frac{1}{{\sqrt V }}$, the value of ${C_P}/{C_V}$ of gas isView Solution
- 6An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 7An ideal gas in a cylinder is separated by a piston in such a way that the entropy of one part is $S_{1}$ and that of the other part is $S_{2}$. Given that $S _{1}> S _{2}$. If the piston is removed then the total entropy of the system will be :View Solution
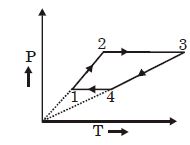
- 8Three moles of an ideal monoatomic gas perform a cycle as shown in the figure. The gas temperature in different states are: $T_1 = 400\, K, T_2 = 800\, K, T_3 = 2400\,K$ and $T_4 = 1200\,K.$ The work done by the gas during the cycle is ........ $kJ$View Solution
- 9One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will beView Solution
- 10A system is provided with $200 \,cal$ of heat and the work done by the system on the surrounding is $40 \,J$. Then its internal energyView Solution
