Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
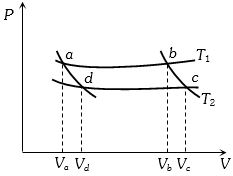
- 2For one complete cycle of a thermodynamic processes on a gas as shown in $P-V$ diagram. Which is trueView Solution
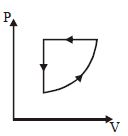
- 3An ideal gas is taken through the cycle $A \to B \to C \to A,$ as shown in the figure. What is the change in internal energy ...... $J$.View Solution
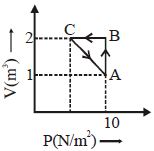
- 4View SolutionThe internal energy of an ideal gas depends upon
- 5In a thermodynamic process two moles of a monatomic ideal gas obeys $P \propto V^{-2}$. If temperature of the gas increases from $300 \,K$ to $400 \,K$, then find work done by the gas .........$R$ (where $R=$ universal gas constant).View Solution
- 6An engine has an efficiency of $1/6.$ When the temperature of sink is reduced by $62^o C,$ its efficiency is doubled. Temperatures of the source is ....... $^oC$View Solution
- 7In a Carnot engine, when ${T_2} = {0^o}C$ and ${T_1} = {200^o}C,$ its efficiency is ${\eta _1}$ and when ${T_1} = 0{\,^o}C$ and ${T_2} = - 200{\,^o}C$, Its efficiency is ${\eta _2}$, then what is ${\eta _1}/{\eta _2}$View Solution
- 8The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 9Which of the accompanying $PV$, diagrams best represents an isothermal processView Solution
- 10View SolutionThe internal energy of an ideal gas increases during an isothermal process when the gas is
