For $a$ gas $C_{p}-C_{V}=R$ in a state $P$ and $C_{p}-C_{V}=1.10 R$ in a state $Q, T_{p}$ and $T_{Q}$ are the temperatures in two different states $P$ and $Q$ respectively. Then
JEE MAIN 2021, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At $27°C$ temperature, the kinetic energy of an ideal gas is ${E_1}$. If the temperature is increased to $327°C,$ then kinetic energy would beView Solution
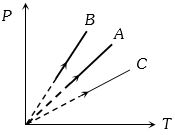
- 2Pressure versus temperature graph of an ideal gas at constant volume $V$ of an ideal gas is shown by the straight line $A$. Now mass of the gas is doubled and the volume is halved, then the corresponding pressure versus temperature graph will be shown by the lineView Solution
- 3View SolutionIt is possible for a substance to coexist in all three phases in equilibrium, when the substance is at
- 4If the pressure of an ideal gas contained in a closed vessel is increased by $0.5\%,$ the increase in temperature is $2K.$ The initial temperature of the gas is ...... $^oC$View Solution
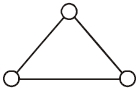
- 5Consider a gas of triatomic molecules. The molecules are assumed to the triangular and made of massless rigid rods whose vertices are occupied by atoms. The internal energy of a mole of the gas at temperature $T$ is $......RT$View Solution
- 6View SolutionWhich of the following plots represents schematically the dependence of the time period of a pendulum, if measured and plotted as a function of the amplitude of its oscillations? (Note amplitude need not be small)
- 7A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$View Solution
- 8$C{O_2}(O - C - O)$ is a triatomic gas. Mean kinetic energy of one gram gas will be (If $N-$Avogadro's number, $k-$Boltzmann's constant and molecular weight of $C{O_2} = 44$)View Solution
- 9The specific heat at constant volume for the monoatomic argon is $0.075\, kcal/kg-K,$ whereas its gram molecular specific heat ${C_V}$ $= 2.98\, cal/mole/K.$ The mass of the argon atom isView Solution
$(Avogadro’s \,number = 6.02 \times {10^{23}}\,molecules/mole)$
- 10View SolutionFor matter to exist simultaneously in gas and liquid phases
