$105$ calories of heat is required to raise the temperature of $3$ moles of an ideal gas at constant pressure from $30^{\circ} C$ to $35^{\circ} C$. The amount of heat required in calories to raise the temperature of the gas through the range $\left(60^{\circ} C\right.$ to $\left.65^{\circ} C \right)$ at constant volume is ........ $cal$ $\left(\gamma=\frac{C_p}{C_v}=1.4\right)$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1On any planet, the presence of atmosphere implies (${C_{rms}}$= root mean square velocity of molecules and ${V_e}$= escape velocity)View Solution
- 2A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 3$\frac{1}{2} $ mole of helium gas is contained in a container at $S.T.P.$ The heat energy needed to double the pressure of the gas, keeping the volume constant (specific heat of the gas $ = 3\,J\,g{m^{ - 1}}\,{K^{ - 1}})$ is ...... $J$View Solution
- 4What is the mass of $2$ litres of nitrogen at $22.4$ atmospheric pressure and $273\,K$View Solution
- 5View SolutionAccording to law of equipartition of energy the molar specific heat of a diatomic gas at constant volume where the molecule has one additional vibrational mode is :-
- 6At a given temperature the ratio of $r.m.s.$ velocities of hydrogen molecule and helium atom will beView Solution
- 7The mean kinetic energy of a gas at $300 K$ is $100 J.$ The mean energy of the gas at $450 K$ is equal to ...... $J$View Solution
- 8The kinetic energy of one gm-mole of a gas at normal temperature and pressure is $(R = 8.31 J/Mole-K)$View Solution
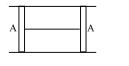
- 9A cylindrical tube of cross-sectional area $A$ has two air tight frictionless pistons at its two ends. The pistons are tied with a straight two ends. The pistons are tied with a straight piece of metallic wire. The tube contains a gas at atmospheric pressure $P_0$ and temperature $T_0$. If temperature of the gas is doubled then the tension in the wire isView Solution
- 10At what temperature $r.m.s.$ speed of air molecules doubles of that at $N.T.P.$ is ...... $^oC$View Solution
