Consider $1 \,kg$ of liquid water undergoing change in phase to water vapour at $100^{\circ} C$. At $100^{\circ} C$, the vapour pressure is $1.01 \times 10^5 \,N - m ^2$ and the latent heat of vaporization is $22.6 \times 10^5 \,Jkg ^{-1}$. The density of liquid water is $10^3 \,kg m ^{-3}$ and that of vapour is $\frac{1}{1.8} \,kg m ^{-3}$. The change in internal energy in this phase change is nearly ............ $\,J kg ^{-1}$
KVPY 2009, Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it. - 2The volume of air increases by $5\%$ in its adiabatic expansion. The percentage decrease in its pressure will be ...... $\%$View Solution
- 3Gas obey $P^2V =$ constant. The initial temperature and volume are $T_0$ and $V_0$. If gas expands to volume $2V_0$, the final temp isView Solution
- 4Two ideal Carnot engines operate in cascade (all heat given up by one engine is used by the other engine to produce work) between temperatures, $\mathrm{T}_{1}$ and $\mathrm{T}_{2} .$ The temperature of the hot reservoir of the first engine is $\mathrm{T}_{1}$ and the temperature of the cold reservoir of the second engine is $\mathrm{T}_{2} . T$ is temperature of the sink of first engine which is also the source for the second engine. How is $T$ related to $\mathrm{T}_{1}$ and $\mathrm{T}_{2}$, if both the engines perform equal amount of work?View Solution
- 5Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
- 6A gas ($\gamma = 1.3)$ is enclosed in an insulated vessel fitted with insulating piston at a pressure of ${10^5}\,N/{m^2}$. On suddenly pressing the piston the volume is reduced to half the initial volume. The final pressure of the gas isView Solution
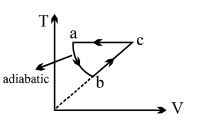
- 7$PV$ curve for the process whose $VT$ curve isView Solution
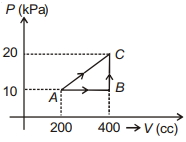
- 8If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
- 9If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 10Given below are two statements:View Solution
Statement $I:$ If heat is added to a system, its temperature must increase.
Statement $II:$ If positive work is done by a system in a thermodynamic process, its volume must increase.
In the light of the above statements, choose the correct answer from the options given below
