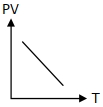
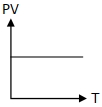
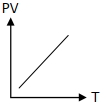
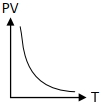
Which of the following graphs represent the behavior of an ideal gas ? Symbols have their usual meaning.
JEE MAIN 2021, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An open and wide glass tube is immersed vertically in mercury in such a way that length $0.05\,\, m$ extends above mercury level. The open end of the tube is closed and the tube is raised further by $0.43 \,\,m$. The length of air column above mercury level in the tube will be ...... $m$ Take $P_{atm} = 76 \,\,cm$ of mercuryView Solution
- 2The equation of state corresponding to $8 \,g$ of ${O_2}$ isView Solution
- 3The number of gas molecules striking per second per square metre of the top surface of a table placed in a room at $20^{\circ} C$ and 1 atmospheric pressure is of the order of $\left(k_{B}=1.4 \times 10^{-23} \,JK ^{-1}\right.$ and the average mass of an air molecule is $5 \times 10^{-27} \,kg$ )View Solution
- 4View SolutionBoyle's law holds for an ideal gas during
- 5A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
- 6If a given mass of gas occupies a volume of $10\, cc$ at $1$ atmospheric pressure and temperature of $100°C(373.15\, K).$ What will be its volume at 4 atmospheric pressure; the temperature being the same .... $cc$View Solution
- 7The correct relation between the degree of freedom $f$ and the ratio of specific heat $\gamma$ isView Solution
- 8The pressure and temperature of two different gases is $P$ and $T$ having the volume $V$ for each. They are mixed keeping the same volume and temperature, the pressure of the mixture will beView Solution
- 9A cylinder of fixed capacity of $44.8 \,litres$ contains helium gas at standard temperature and pressure. The amount of heat needed to raise the temperature of gas in the cylinder by $20.0^{\circ} C$ will be .............. $J$(Given gas constant $R =8.3 \,JK ^{-1}- moI ^{-1}$ )View Solution
- 10When $2 \,\,gms$ of a gas are introduced into an evacuated flask kept at $25\,^0C$ the pressure is found to be one atmosphere. If $3 \,\,gms$ of another gas added to the same flask the pressure becomes $1.5$ atmospheres. The ratio of the molecular weights of these gases will beView Solution