A vessel of volume $0.02 \,\,m^3$ contains a mixture of hydrogen and helium at $20\,^oC$ and $2$ atmospheric pressure. The mass of mixture is $5\,\, gms$. Find the ratio of mass of hydrogen to that of helium in the mixture.
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $\mathrm{n}$ is the number density and $\mathrm{d}$ is the diameter of the molecule, then the average distance covered by a molecule between two successive collisions (i.e. mean free path) is represented by :View Solution
- 2$\mathrm{N}$ moles of a polyatomic gas $(f=6)$ must be mixed with two moles of a monoatomic gas so that the mixture behaves as a diatomic gas. The value of $\mathrm{N}$ is:View Solution
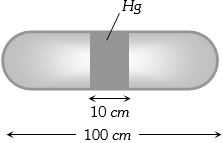
- 3A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 4$Assertion$: The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5\, times$ the product of its pressure and its volume.View Solution
$Reason$ : The molecules of a gas collide with each other and the velocities of the molecules change due to the collision. - 5When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
- 6The value of $PV/T$ for one mole of an ideal gas is nearly equal to ......... $J\, mol^{-1}K^{-1}$View Solution
- 7A monatomic ideal gas undergoes a process in which the ratio of $\mathrm{P}$ to $\mathrm{V}$ at any instant is constant and equals to $1$. What is the molar heat capacity of the gas?View Solution
- 8If a mixture of $28\,\, g$ of Nitrogen, $4 \,\,g$ of Hydrogen and $8 \,\,gm$ of Helium is contained in a vessel at temperature $400 \,\,K$ and pressure $8.3 \times 10^5 \,\,Pa$, the density of the mixture will be :View Solution
- 9According to $C.E.$ van der Waal, the interatomic potential varies with the average interatomic distance $(R)$ asView Solution
- 10View SolutionVelocity of sound measured at a given temperature in oxygen and hydrogen is in the ratio
