Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
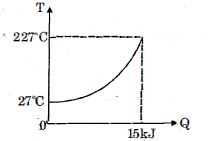
- 1$Q$ amount of heat is given to $0.5\ mole$ of an ide al mono-atomic gas by a process $TV^n$ constant. Following graph shows variation of temperature with $Q$ . Find value of $n$.View Solution
- 2View SolutionWhen an ideal triatomic non-linear gas is heated at constant pressure, the fraction of the heat energy supplied which increases the internal energy of the gas is
- 3A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
- 4View SolutionThe temperature of an ideal gas is kept constant as it expands. The gas does external work. During this process, the internal energy of the gas
- 5View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally
- 6A Carnot engine, whose efficiency is $40\%$, takes in heat from a source maintained at a temperature of $500\ K$. It is desired to have an engine of efficiency $60\%$. Then, the intake temperature for the same exhaust (sink) temperature must be ....... $K$View Solution
- 7A Container having $1$ mole of a gas at a temperature $27°C$ has a movable piston which maintains at constant pressure in container of $1 \,atm.$ The gas is compressed until temperature becomes $127°C$. The work done is ........ $J$ ($C_P$ for gas is $7.03\, cal/mol-K)$View Solution
- 8View SolutionWhich of the following is a slow process
- 9When heat energy of $1500\; Joules$, is supplied to a gas at constant pressure $2.1 \times {10^5}\;N/{m^2}$, there was an increase in its volume equal to $2.5 \times {10^{ - 3}}\;{m^3}$. The increase in internal energy of the gas in Joules is ...... $J$View Solution
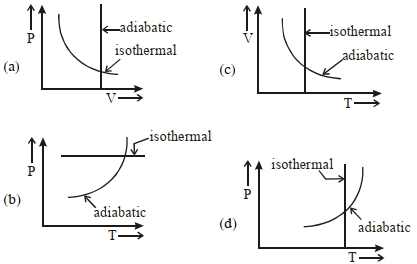
- 10View SolutionWhich one is the correct option for the two different thermodynamic processes ?