Heat is given to an ideal gas in an isothermal process.
$A.$ Internal energy of the gas will decrease.
$B.$ Internal energy of the gas will increase.
$C.$ Internal energy of the gas will not change.
$D.$ The gas will do positive work.
$E.$ The gas will do negative work.
Choose the correct answer from the options given below :
JEE MAIN 2023, Medium
$dQ = dU + dW \Rightarrow dU = nC _{ V } dT$
$dU =0 \quad \text { (for isothermal) }$
$U =\text { constant }$
$Also dQ > 0 \quad \text { (supplied) }$
Hence,$dW > 0$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhen a gas expands adiabatically
- 2$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
- 3View SolutionWhich of the following is not a thermodynamics co-ordinate
- 4A van der Waal's gas obeys the equation of state $\left(p+\frac{n^2 a}{V^2}\right)(V-n b)=n R T$. Its internal energy is given by $U=C T-\frac{n^2 a}{V}$. The equation of a quasistatic adiabat for this gas is given byView Solution
- 5$1 \,\,kg$ of a gas does $20\,\, kJ$ of work and receives $16 \,\,kJ$ of heat when it is expanded between two states. $A$ second kind of expansion can be found between the initial and final state which requires a heat input of $9\,\, kJ$. The work done by the gas in the second expansion is ....... $kJ$View Solution
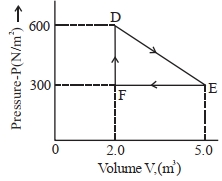
- 6A thermodynamic system is taken from an original state $D$ to an intermediate state $E$ by the linear process shown in the figure. Its volume is then reduced to the original volume from $E$ to $F$ by an isobaric process. The total work done by the gas from $D$ to $E$ to $F$ will be $......J$View Solution
- 7An electric appliance supplies $6000\, {J} / {min}$ heat to the system. If the system delivers a power of $90\, {W}$. How long (in $sec$) it would take to increase the internal energy by $2.5 \times 10^{3}\, {J}$ ?View Solution
- 8View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
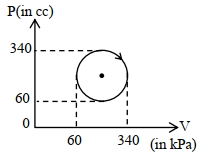
- 9View SolutionThe heat absorbed by a system in going through the given cyclic process is:
- 10If $\gamma $ denotes the ratio of two specific heats of a gas, the ratio of slopes of adiabatic and isothermal $PV$ curves at their point of intersection isView Solution
