Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The specific heat capacity of a metal at low temperature $(T)$ is given as $C_p=32\left(\frac{ T }{400}\right)^{3}\;kJ\,k ^{-1}\, kg ^{-1}$. A $100\; g$ vessel of this metal is to be cooled from $20 \;K$ to $4\; K$ by a special refrigerator operating at room temperature $27^\circ c$). The amount of work required to cool the vessel isView Solution
- 2View SolutionIn the cyclic process shown in the figure, the work done by the gas in one cycle is
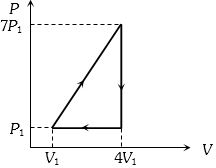
- 3View SolutionA measure of the degree of disorder of a system is known as
- 4View SolutionThe given diagram shows four processes i.e., isochoric, isobaric, isothermal and adiabatic. The correct assignment of the processes, in the same order is given by
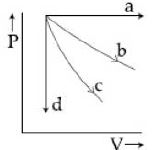
- 5In a given process for an ideal gas, $dW = 0$ and $dQ < 0.$ Then for the gasView Solution
- 6In the $P-V$ diagram, $I$ is the initial state and $F$ is the final state. The gas goes from $I$ to $F$ by $(i)\,IAF,\,\,(ii)\,IBF,\,\,(iii) ICF$. The heat absorbed by the gas isView Solution
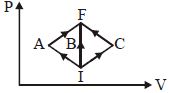
- 7One mole of a monatomic ideal gas is taken through a cycle $ABCDA$ as shown in the $P-V$ diagram. Column $II$ gives the characteristics involved in the cycle. Match them with each of the processes qiven in Column $I$View Solution
Column $I$ Column $II$ $(A)$ Process $A \rightarrow B$ $(p)$ Internal energy decreases. $(B)$ Process $B \rightarrow C$ $(q)$ Internal energy increases. $(C)$ Process $C \rightarrow D$ $(r)$ Heat is lost. $(D)$ Process $D \rightarrow A$ $(s)$ Heat is gained. $(t)$ Work is done on the gas. 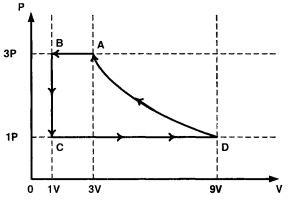
- 8Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. The net work done on the gas in the cycle $ABCDA$ is ...... $R$View Solution

- 9One mole of an ideal gas is contained with in a cylinder by a frictionless piston and is initially at temperature $T$. The pressure of the gas is kept constant while it is heated and its volume doubles. If $R$ is molar gas constant, the work done by the gas in increasing its volume isView Solution
- 10One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
