In the following figure, four curves $A, B, C$ and $D$ are shown. The curves are
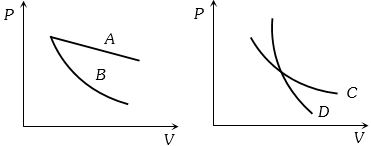

Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
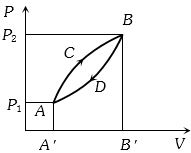
- 1A thermodynamic system is taken from state $A$ to $B$ along $ACB$ and is brought back to $A$ along $BDA$ as shown in the $PV$ diagram. The net work done during the complete cycle is given by the areaView Solution
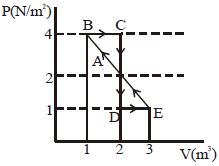
- 2One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
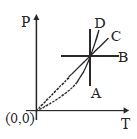
- 3Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternativeView Solution
- 4Two Carnot engines $A$ and $B$ are operated in series. Engine $A$ receives heat from a reservoir at $600\,K$ and rejects heat to a reservoir at temperature $T$. Engine $B$ receives; heat rejected by engine $A$ and in turn rejects it to a reservoir at $100\,K$. If the efficiencies of the two engines $A$ and $B$ are represented by ${\eta _A}$ and ${\eta _B}$ respectively, then what is the value of $\frac{{{\eta _A}}}{{{\eta _B}}}$View Solution
- 5Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
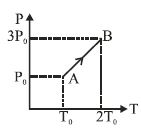
- 6The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution
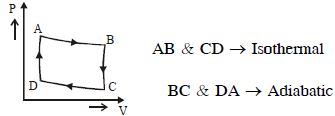
- 7A monoatomic gas of $n-$moles is heated from temperature $T_1$ to $T_2$ under two different conditions $(i)$ at constant volume and $(ii)$ at constant pressure. The change in internal energy of the gas isView Solution
- 8A refrigerator works between $4^o C$ and $30^o C.$ It is required to remove $600$ calories of heat every second in order to keep the temperature of the refrigerated space constant. The power required is ....... $W$ (Take $1\, cal \,=\, 4.2\, Joules\,)$View Solution
- 9View SolutionWhich of the following process will give maximum amount of heat to surrounding when volume becomes half of initial
- 10A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is $3$ times the amount of work done by the cycle, the efficiency of the cycle isView Solution
