Ideal mono-atomic gas is taken through process such that $dQ= 3dU.$ The molar heat capacity for this process is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The temperature of an ideal gas is increased from $200\,K$ to $800\,K$. If r.m.s. speed of gas at $200\,K$ is $v_0$. Then, r.m.s. speed of the gas at $800\,K$ will be:View Solution
- 2If pressure of $C{O_2}$ (real gas) in a container is given by $P = \frac{{RT}}{{2V - b}} - \frac{a}{{4{b^2}}}$ then mass of the gas in container is ...... $gm$View Solution
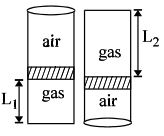
- 3An ideal gas is trapped inside a test tube of cross-sectional area $20 \times 10^{-6} \,\,m^2$ as shown in the figure. The gas occupies a height $L_1$ at the bottom of the tube and is separated from air at atmospheric pressure by a mercury column of mass $0.002\,\, kg$. If the tube is quickly turned isothermally, upside down so that $L_2$ mercury column encloses the gas from below. The gas now occupies height $L_1$ in the tube. The ratio $L_1$ is [Take atmospheric pressure $= 10^5 Nm^{-2}]$View Solution
- 4Five gas molecules chosen at random are found to have speeds of $500,600,700,800$ and $900 m / s$. Then which of the following statements is correct?View Solution
- 5At $NTP,$ sample of equal volume of chlorine and oxygen is taken. Now ratio of No. of moleculesView Solution
- 6If a given mass of gas occupies a volume of $10\, cc$ at $1$ atmospheric pressure and temperature of $100°C(373.15\, K).$ What will be its volume at 4 atmospheric pressure; the temperature being the same .... $cc$View Solution
- 7A certain amount of an ideal monatomic gas needs $20 \,J$ of heat energy to raise its temperature by $10^{\circ} C$ at constant pressure. The heat needed for the same temperature rise at constant volume will be ........ $J$View Solution
- 8The total kinetic energy of translatory motion of all the molecules of $5$ litres of nitrogen exerting a pressure $P$ is $3000 \,\,J$.View Solution
- 9At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
- 10Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)View Solution
