Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Suppose ideal gas equation follows $V{P^3}$= constant. Initial temperature and volume of the gas are $T$ and $V$ respectively. If gas expand to $27V$ then its temperature will be comeView Solution
- 2View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 3A gas mixture consists of $8$ moles of argon and $6$ moles of oxygen at temperature $T$. Neglecting all vibrational modes, the total internal energy of the system isView Solution
- 4Water falls from a height of $60\,m$ at the rate of $15\,kg/s$ to operate a turbine. The losses due to frictional forces are $10\%$ of energy. How much power is generated by the turbine ....... $kW$ $(g = 10\,m/s^2)$View Solution
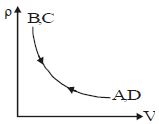
- 5Density $vs$ volume graph is shown in the figure. Find corresponding pressure $vs$ temperature graphView Solution
- 6A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
- 7An ideal gas at atmospheric pressure is adiabatically compressed so that its density becomes $32$ times of its initial value. If the final pressure of gas is $128$ atmosphers, the value of $\gamma$ the gas isView Solution
- 8View SolutionFor a reversible process, necessary condition is
- 9In a thermodynamics process, pressure of a fixed mass of a gas is changed in such a manner that the gas releases $20 J$ of heat and $8J$ of work is done on the gas. If the initial internal energy of the gas was $30J.$ The final internal energy will be ...... $J$View Solution
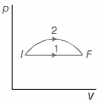
- 10An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,View Solution