If $C_p$ and $C_v$ denote the specific heats of nitrogen per unit mass at constant pressure and constant volume respectively, then
AIEEE 2007, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Let $\bar v,\;{v_{rms}}$ and ${v_p}$ respectively denote the mean speed, root mean square speed and most probable speed of the molecules in an ideal monoatomic gas at absolute temperature $T.$ The mass of a molecule is $m.$ ThenView Solution
- 2A system consists of two types of gas molecules $A$ and $B$ having same number density $2 \times$ $10^{25}\, / {m}^{3}$. The diameter of ${A}$ and ${B}$ are $10\, \stackrel{\circ}{{A}}$ and $5\, \stackrel{\circ}{{A}}$ respectively. They suffer collision at room temperature. The ratio of average distance covererd by the molecule $A$ to that of $B$ between two successive collision is $.....\,\times 10^{-2}$View Solution
- 3Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent degree of freedom $f$ of the mixture isView Solution
- 4Air is filled at $60^o C$ in a vessel of open mouth. The vessel is heated to a temperature $T$ so that $1/4^{th}$ part of air escapes. Assuming the volume of the vessel remaining constant, the value of $T$ is ....... $^oC$View Solution
- 5If two moles of diatomic gas and one mole of mono atomic gas are mixed then the ratio of specific heats $\gamma=$ ......View Solution
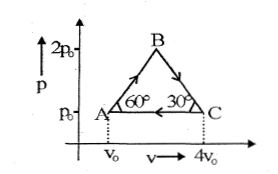
- 6Four moles of an ideal monoatomic gas undergoes a cyclic process $ABCA$ as shown in figure find ratio of temperature of $A$ and $B$ .View Solution
- 7A thermally insulated vessel contains an ideal gas of molecular mass $M$ and ratio of specific heats $1.4$. Vessel is moving with speed $v$ and is suddenly brought to rest. Assuming no heat is lost to the surrounding and vessel temperature of the gas increases by ... ( $R =$ universal gas constant )View Solution
- 8In Maxwell's speed distribution curve, for $N_2$ gas, the average of $\mid$ relative velocity (in $m/s$) $\mid$ between two molecules at $300 \,K$ will beView Solution
- 9Relation between $U, P$ and $V$ for ideal gas is $U= 2+2PV$ then gas isView Solution
- 10The change in the magnitude of the volume of an ideal gas when a small additional pressure $\Delta P$ is applied at a constant temperature, is the same as the change when the temperature is reduced by a small quantity $\Delta T$ at constant pressure. The initial temperature and pressure of the gas were $300\, K$ and $2\; atm$ respectively. If $|\Delta T|=C|\Delta P|$ then value of $C$ in $(K / a t m)$ is......View Solution
