One mole of an ideal monoatomic gas is compressed isothermally in a rigid vessel to double its pressure at room temperature, $27\,^oC$.The work done on the gas will be
JEE MAIN 2018, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionWhen an ideal monoatomic gas is heated at constant pressure, fraction of heat energy supplied which increases the internal energy of gas, is
- 2Find the amount of work done to increase the temperature of one mole of an ideal gas by $30^o\ C$ if it is expanding under the condition .... $J$View Solution
$V\propto {T^{\frac{2}{3}}}$ $[R = 1.99\ cal/mol-K]$
- 3One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
- 4A monoatomic gas $\left( {\gamma = \frac{5}{3}} \right)$ is suddenly compressed to $\frac{1}{8}$ of its original volume, then the pressure of gas will change to how many times the initial pressure?View Solution
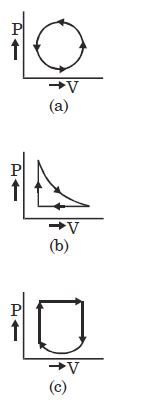
- 5Work done for the process shown in the figure is ............ $J$View Solution
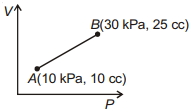
- 6One mole of ${O_2}$ gas having a volume equal to $22.4$ litres at ${0^o}C$ and $1$ atmospheric pressure in compressed isothermally so that its volume reduces to $11.2$ litres. The work done in this process is ...... $J$View Solution
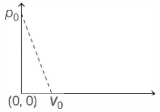
- 7One mole of ideal gas undergoes a linear process as shown in the figure below. Its temperature expressed as a function of volume $V$ isView Solution
- 8One mole of an ideal gas goes from an initial state $A$ to final state $B$ via two processes : It first undergoes isothermal expansion from volume $V$ to $3\, V$ and then its volume is reduced from $3\, V$ to $V$ at constant pressure. The correct $P-V$ diagram representing the two processes isView Solution
- 9$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 10View SolutionWhat is the nature of change in internal energy in the following three thermodynamical processes shown in figure