The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)
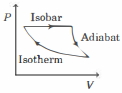

KVPY 2020, Advanced
$(b)$ The given $p-V$ diagram can be shown as,
For curve $A B$ (isobaric),
$\frac{V_{B}-V_{A}}{T_{B}-T_{A}}$ ...........$(i)$
For curve $B C$ (adiabatic),
$T_{B}^{\top} V_{B}^{\gamma-1} =T_{C} V_{C}^{\gamma-1}$ ..........$(ii)$
$\text { Since, } V_{B} =x V_{A}$
$\text { and } V_{C} =x\left(\frac{\gamma}{\gamma-1}\right) V_{A}$
Efficiency of cycle,
$\eta=1-\frac{Q_{C A}}{Q_{A B}}$
$=1-\frac{n R T_{A} \ln \left(\frac{V_{C}}{V_{A}}\right)}{\frac{n \gamma}{\gamma-1)}\left(T_{B}-T_{A}\right)}$
$=1-\frac{\left(\frac{\gamma}{\gamma-1}\right) \ln x}{\left(\frac{\gamma}{\gamma-1}\right)(x-1)} \quad\left[\because \frac{T_{B}}{T_{A}}=x\right]$
Given, $\eta=50 \%=\frac{1}{2}$
$\Rightarrow \quad \frac{1}{2}=\frac{\ln x}{x-1} \Rightarrow x^{2}=e^{x-1}$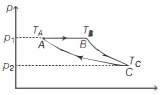
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Find the amount of work done to increase the temperature of one mole of an ideal gas by $30^o\ C$ if it is expanding under the condition .... $J$View Solution
$V\propto {T^{\frac{2}{3}}}$ $[R = 1.99\ cal/mol-K]$
- 2Two cylinders $A$ and $B$ fitted with pistons contain equal amounts of an ideal diatomic gas at $300 K$ . The piston of $A$ is free to move while that of $B$ is held fixed. The same amount of heat is given to the gas in each cylinder. If the rise in temperature of the gas in $A$ is $30 K$ , then the rise in temperature of the gas in $B$ is ..... $K$View Solution
- 3View SolutionThe rate of recombination or generation are governed by the law(s) of
- 4The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 5A diatomic gas with rigid molecules does $10\, J$ of work when expanded at constant pressure. What would be the heat energy absorbed by the gas, in this process ..... $J$.View Solution
- 6The latent heat of vaporisation of water is $2240\, J/gm$. If the work done in the process of expansion of $1 \,g$ is $168 \,J$, then increase in internal energy is ....... $J$View Solution
- 7Consider two containers $A$ and $B$ containing monoatomic gases at the same Pressure $(P)$, Volume $(V)$ and Temperature $(T)$. The gas in $A$ is compressed isothermally to $\frac{1}{8}$ of its original volume while the gas $B$ is compressed adiabatically to $\frac{1}{8}$ of its original volume. The ratio of final pressure of gas in $B$ to that of gas in $A$ is ...........View Solution
- 8The efficiency of a Carnot engine operating with reservoir temperature of $100\,^{\circ} C$ and $-23\,^{\circ} C$ will beView Solution
- 9A gas is taken through the cycle $A\to B\to C\to A$ as shown. What is the net work done by the gas ...... $J$ $?$View Solution
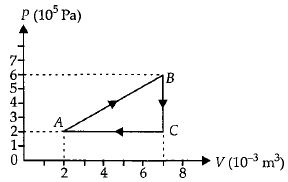
- 10A Carnot's engine works as a refrigerator between $250\, K$ and $300\, K$. It receives $500\, cal$ heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is ..... $J$View Solution
