If heat is added at constant volume, $6300\,\,J$ of heat are required to raise the temperature of an ideal gas by $150\,\,K$. If instead, heat is added at constant pressure, $8800$ joules are required for the same temperature change. When the temperature of the gas changes by $300\,\,K$, the internal energy of the gas changes by ..... $J$
Diffcult
Heat added at constant volume is equal to the change in internal energy of the system. Change in internal energy $\quad \Delta U=n C_{v} \Delta T$
For $\Delta T=150 K, \quad \Delta U=Q_{v}=6300 \mathrm{J}$
$\therefore \quad 6300=n C_{v}(150)$ $\ldots(1)$
Let the change in internal energy be $\Delta U$ for $\Delta T=300 K$
$\therefore \Delta U=n C_{v}(300)$ $...(2)$
Dividing $( 2 )$ and $( 1 )$ we get $\frac{\Delta U}{6300}=\frac{300}{150}$
$\Longrightarrow \Delta U=12600 \mathrm{J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In a process, temperature and volume of one mole of an ideal monoatomic gas are varied according to the relation $VT = K$, where $I$ is a constant. In this process the temperature of the gas is increased by $\Delta T$. The amount of heat absorbed by gas is ($R$ is gas constant)View Solution
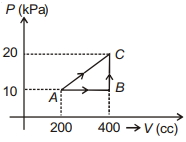
- 2If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
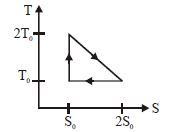
- 3View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
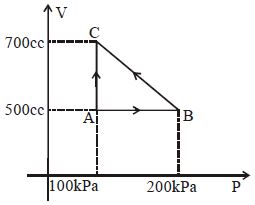
- 4A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
- 5A Carnot engine has efficiency $25\%$ . It operates between reservoirs of constant temperature with temperature difference of $80\,K$ . What is the temperature of low temperature reservoir ...... $^oC$View Solution
- 6View SolutionDuring an isothermal expansion of an ideal gas
- 7View SolutionFor a reversible process, necessary condition is
- 8One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
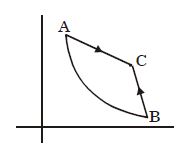
- 9Graph $A-B$ is an adiabatic curve. Choose the correct statementView Solution
- 10View SolutionIn adiabatic expansion
