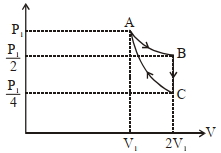
$W _{ AB }= P _{1} V _{1} \ln \left[\frac{2 V _{1}}{ V _{1}}\right]= P _{1} V _{1} \ln (2)$
$B - C \rightarrow$ Isochoric process
$W _{ BC }=0$
$C - A \rightarrow$ Adiabatic process
$W _{ CA }=\frac{ P _{1} V _{1}-\frac{ P _{1}}{4} \times 2 V _{1}}{1-\gamma}=\frac{ P _{1} V _{1}\left[1-\frac{1}{2}\right]}{1-\gamma}=\frac{ P _{1} V _{1}}{2(1-\gamma)}$
$W _{ net }= W _{ AB }+ W _{ BC }+ W _{ CA } \quad\left\{ P _{1} V _{1}= RT \right\}$
$= P _{1} V _{1} \ln (2)+0+\frac{ P _{1} V _{1}}{2(1-\gamma)}$
$W _{\text {net }}= RT \left[\ln (2)-\frac{1}{2(\gamma-1)}\right]$
Download our appand get started for free
Similar Questions
- 1View SolutionIrreversible process is
- 2During an isothermal expansion, a confined ideal gas does $-150 \,J$ of work against its surroundings. This implies thatView Solution
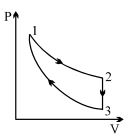
- 3Three processes form a thermodynamic cycle as shown on $P-V$ diagram for an ideal gas. Process $1 \rightarrow 2$ takes place at constant temperature $(300K$). Process $2 \rightarrow 3$ takes place at constant volume. During this process $40J$ of heat leaves the system. Process $3 \rightarrow 1$ is adiabatic and temperature $T_3$ is $275K$. Work done by the gas during the process $3 \rightarrow 1$ is ..... $J$View Solution
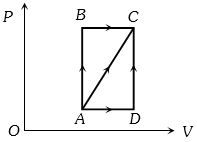
- 4A thermodynamic process is shown in the figure. The pressures and volumes corresponding to some points in the figure are :View Solution
${P_A} = 3 \times {10^4}Pa,\;{P_B} = 8 \times {10^4}Pa$ and ${V_A} = 2 \times {10^{ - 3}}{m^3},\;{V_D} = 5 \times {10^{ - 3}}{m^3}$
In process $AB$, $600 J$ of heat is added to the system and in process $BC, 200 J $ of heat is added to the system. The change in internal energy of the system in process $ AC$ would be ...... $J$
- 5$2$ moles of a monoatomic gas are expanded to double its initial volume, through a process $P/V =$ constant. If its initial temperature is $300\,\, K$, then which of the following is not true.View Solution
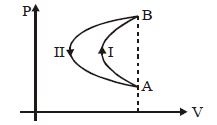
- 6In a cyclic process, a gas is taken from state $A$ to $B$ via path $-I$ as shown in the indicator diagram and taken back to state $A$ from state $B$ via path $-II$ . In the complete cycleView Solution
- 7A thermally isolated cylindrical closed vessel of height $8 m$ is kept vertically. It is divided into two equal parts by a diathermic $($perfect thermal conductor$)$ frictionless partition of mass $8.3 kg$. Thus the partition is held initially at a distance of $4 m$ from the top, as shown in the schematic figure below. Each of the two parts of the vessel contains $0.1$ mole of an ideal gas at temperature $300 K$. The partition is now released and moves without any gas leaking from one part of the vessel to the other. When equilibrium is reached, the distance of the partition from the top $($in $m )$ will be $. . . . . ($take the acceleration due to gravity $=10 ms ^{-2}$ and the universal gas constant $=8.3 J mol ^{-1} K ^{-1} ).$View Solution
- 8One mole of a monatomic ideal gas undergoes the cyclic process $J \rightarrow K \rightarrow L \rightarrow M \rightarrow J$, as shown in the $P - T$ diagram.View Solution
Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
$List-I$ $List-II$ ($P$) Work done in the complete cyclic process ($1$) $R T_0-4 \ R T_0 \ln 2$ ($Q$) Change in the internal energy of the gas in the process $JK$ ($2$) $0$ ($R$) Heat given to the gas in the process $KL$ ($3$) $3 \ R T_0$ ($S$) Change in the internal energy of the gas in the process $MJ$ ($4$) $-2 \ R T_0 \ln 2$ ($5$) $-3 \ R T_0 \ln 2$ 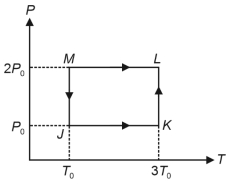
- 9A carnot engine is made to work between $200\,^oC$ and $0\,^oC$ first and then between $0\,^oC$ and $-200\,^oC$. The ratio of efficiencies $\left( {\frac{{{\eta _2}}}{{{\eta _1}}}} \right)$ of the engine in two cases isView Solution
- 10View SolutionWhich of the following processes is reversible
