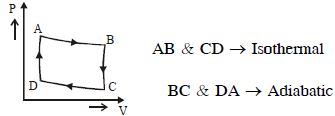Match the quantities mentioned in $List-I$ with their values in $List-II$ and choose the correct option. [ $R$ is the gas constant]
| $List-I$ | $List-II$ |
| ($P$) Work done in the complete cyclic process | ($1$) $R T_0-4 \ R T_0 \ln 2$ |
| ($Q$) Change in the internal energy of the gas in the process $JK$ | ($2$) $0$ |
| ($R$) Heat given to the gas in the process $KL$ | ($3$) $3 \ R T_0$ |
| ($S$) Change in the internal energy of the gas in the process $MJ$ | ($4$) $-2 \ R T_0 \ln 2$ |
| ($5$) $-3 \ R T_0 \ln 2$ |
$K \left( P _0, 3 V _0, 3 T _0\right)$
$M \left(2 P _0, \frac{ V _0}{2}, T _0\right)$
$L \left(2 P _0, \frac{3 V _0}{2}, 3 T _0\right)$
$P _0 V _0= nRT _0$
$JK \rightarrow \text { isobaric } \Rightarrow W = P _0\left(2 V _0\right)=2 nRT \text {. }$
$\Delta U =\frac{3}{2} nR \left(2 T _0\right)=3 nRT _0$
$KL \rightarrow \text { isothermal } \rightarrow W = nR (3 T ) \ln \left(\frac{1}{2}\right)=-3 nRT _0 \ln 2$
$\Delta U =0 \Rightarrow Q =-3 nRT _0 \ln 2$
$LM \rightarrow \text { isobaric }=2 P _0\left(- V _0\right)=-2 nRT _0$
$MJ \rightarrow \text { isothermal } \Rightarrow nRT _0 \ell n 2 ; \Delta U =0$
$WD _{ xat }=-2 nRT _0 \ln 2$
$P \rightarrow 4, Q \rightarrow 3, R \rightarrow 5, S \rightarrow 2$
Download our appand get started for free
Similar Questions
- 1One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $8.0 \,m ^{3}$ to $1.0 \,m ^{3}$.
Step $2$ Then expanded isothermally at temperature $T_{1}$ to volume $10.0 \,m ^{3}$.
Step $3$ Then expanded adiabatically to volume $80.0 \,m ^{3}$.
Step $4$ Then compressed isothermally at temperature $T_{2}$ to volume $8.0 \,m ^{3}$.
Then, $T_{1} / T_{2}$ is
- 2A hypothetical gas expands adiabatically such that its volume changes from $8$ litres to $27$ litres. If the ratio of final pressure of the gas to initial pressure of the gas is $\frac{16}{81}$. Then the ratio of $\frac{C_P}{C_V}$ will beView Solution
- 3An ideal gas is taken through a quasi-static process described by $P = \alpha\, V^2$, with $\alpha = 5\,atm/m^6$. The gas is expanded to twice its original volume of $1\,m^3$. How much work is done by the expanding gas in this processView Solution
- 4During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
- 5The volume of $1\; mole$ of an ideal gas with the adiabatic exponent $\gamma$ is changed according to the relation $V=\frac bT$ where $b =$ constant. The amount of heat absorbed by the gas in the process if the temperature is increased by $\triangle T$ will beView Solution
- 6If $R =$ universal gas constant, the amount of heat needed to raise the temperature of $2$ mole of an ideal monoatomic gas from $273K$ to $373K$ when no work is done ...... $R$View Solution
- 7An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1 g/sec$. The calorific value of the fuel is $2 kcal/g$. Is the claim of the engineerView Solution
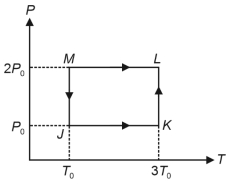
- 8Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
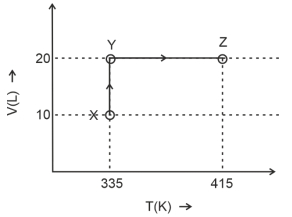
- 9The $P-V$ diagram of $2\,g$ of helium gas for a certain process $A$ $\to$ $B$ is shown in the figure. What is the heat given to the gas during the process $A$ $\to$ $B$?View Solution
- 10The $P-V$ diagram of a certain process (carnot cycle) is as shown. The process is also represent asView Solution