A certain amount of gas of volume $V$ at $27^{o}\,C$ temperature and pressure $2 \times 10^{7} \;Nm ^{-2}$ expands isothermally until its volume gets doubled. Later it expands adiabatically until its volume gets redoubled. The final pressure of the gas will be (Use $\gamma=1.5$ )
JEE MAIN 2022, Medium
$P _{1}=2 \times 10^{7} Pa$
$P _{1} V _{1}= P _{2} V _{2}$
Since $V_{2}=2 V_{1}$ Hence $P_{2}=P_{1} / 2$ (isothermal expansion)
$P _{2}=1 \times 10^{7} Pa$
$P _{2}\left( V _{2}\right)^{\gamma}= P _{3}\left(2 V _{2}\right)^{\gamma}$
$P _{3}=\frac{1 \times 10^{7}}{2^{1.5}}=3.536 \times 10^{6}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
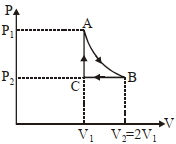
- 1$n$ mole a perfect gas undergoes a cyclic process $ABCA$ (see figure) consisting of the following processes.View Solution
$A \rightarrow B :$ Isothermal expansion at temperature $T$ so that the volume is doubled from $V _{1}$ to $V _{2}=2 V _{1}$ and pressure changes from $P _{1}$ to $P _{2}$
$B \rightarrow C :$ Isobaric compression at pressure $P _{2}$ to initial volume $V _{1}$
$C \rightarrow A$ : Isochoric change leading to change of pressure from $P _{2}$ to $P _{1}$
Total workdone in the complete cycle $ABCA$ is
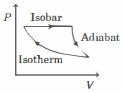
- 2The efficiency of the cycle shown below in the figure (consisting of one isobar, one adiabat and one isotherm) is $50 \%$. The ratio $x$, between the highest and lowest temperatures attained in this cycle obeys (the working substance is an ideal gas)View Solution
- 3A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
- 4A cylinder of mass $1\,kg$ is given heat of $20000\, J$ at atmospheric pressure. If initially temperature of cylinder is $20\,^oC$, then work done by the cylinder will be .......$J$ (Given that Specific heat of cylinder $= 400 \,J\, kg^{-1}$, Coefficient of volume expansion $= 9 \times {10^{-5}}\,^o C^{-1}$, Atmospheric pressure $= 10^5 \,N/m^2$ and density of cylinder $9000\,kg/m^3$)View Solution
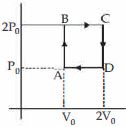
- 5Helium gas goes through a cycle $ABCDA$ ( consisting of two isochoric and isobaric lines) as shown in figure Efficiency of this cycle is nearly ....... $\%$ (Assume the gas to be close to ideal gas)View Solution
- 6If $\Delta Q$ and $\Delta W$ represent the heat supplied to the system and the work done on the system respectively, then the first law of thermodynamics can be written asView Solution
- 7$Assertion :$ When a bottle of cold carbonated drink is opened, a slight fog forms around the opening.View Solution
$Reason :$ Adiabatic expansion of the gas causes lowering of temperature and condensation of water vapours. - 8A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) isView Solution
- 9At $N.T.P.$ one mole of diatomic gas is compressed adiabatically to half of its volume $\gamma = 1.41$. The work done on gas will be ....... $J$View Solution
- 10The amount of work done in an adiabatic expansion from temperature $T$ to ${T_1}$ isView Solution
