The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$
JEE MAIN 2021, Medium
Pressure is not changing $\Rightarrow$ isobaric process
$\Rightarrow \Delta {U}={n} {C}_{{v}} \Delta {T}=\frac{5 {nR} \Delta {T}}{2}$
and ${W}={n} {R} \Delta {T}$
$\frac{\Delta {U}}{{W}}=\frac{5}{2}=\frac{{x}}{10} \Rightarrow {x}=25.00$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The $P-V$ diagram for an ideal gas in a piston cylinder assembly undergoing a thermodynamic process is shown in the figure. The process isView Solution
- 2An engine is supposed to operate between two reservoirs at temperature $727°C$ and $227°C.$ The maximum possible efficiency of such an engine isView Solution
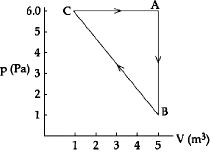
- 3For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution
- 4Two moles of an ideal monoatomic gas at ${27^o}C$ occupies a volume of $V.$ If the gas is expanded adiabatically to the volume $2V,$ then the work done by the gas will be ....... $J$ $[\gamma = 5/3,\,R = 8.31J/mol\,K]$View Solution
- 5A gas expands under constant pressure $P$ from volume ${V_1}$ to ${V_2}$. The work done by the gas isView Solution
- 6One mole of an ideal gas goes from an initial state $A$ to final state $B$ via two processes : It first undergoes isothermal expansion from volume $V$ to $3\, V$ and then its volume is reduced from $3\, V$ to $V$ at constant pressure. The correct $P-V$ diagram representing the two processes isView Solution
- 7View SolutionWhich of the following is a slow process
- 8An ideal gas undergoes a polytropic given by equation $P V^n=$ constant. If molar heat capacity of gas during this process is arithmetic mean of its molar heat capacity at constant pressure and constant volume then value of $n$ is ..............View Solution
- 9An ideal system can be brought from state $A$ to $B$ through four paths as shown in the figure. The energy given to the system is minimum inView Solution
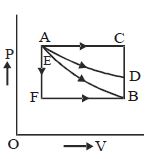
- 10For $P-V$ diagram of a thermodynamic cycle as shown in figure, process $B C$ and $D A$ are isothermal. Which of the corresponding graphs is correct?View Solution