$\mathrm{N}$ moles of a polyatomic gas $(f=6)$ must be mixed with two moles of a monoatomic gas so that the mixture behaves as a diatomic gas. The value of $\mathrm{N}$ is:
JEE MAIN 2024, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The diameter of oxygen molecule is $2.94 \times {10^{ - 10}}m.$ The Vander Waal’s gas constant ‘$b’$ in ${m^3}/mol$ will beView Solution
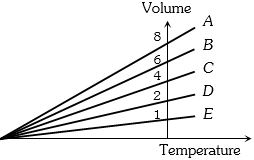
- 2The expansion of an ideal gas of mass $m$ at a constant pressure $P$ is given by the straight line $D$. Then the expansion of the same ideal gas of mass $2m$ at a pressure $P/ 2 $ is given by the straight lineView Solution
- 3The average kinetic energy of a helium atom at ${30^o}C$ isView Solution
- 4A partition divides a container having insulated walls into two compartments $I$ and $II$. the same gas fills the two compartments. The ratio of the number of molecules in compartments $I$ and $II$ isView Solution

- 5A vessel contains $14\,g$ of nitrogen gas at a temperature of $27^{\circ}\,C$. The amount of heat to be transferred to the gap to double the r.m.s. speed of its molecules will be $......J$ $\left(\right.$ Take $R =8.32\,J\,mol ^{-1} k ^{-1}$ )View Solution
- 6If the $rms$ speed of oxygen molecules at $0^{\circ} {C}$ is $160\; {m} / {s}$, find the rms speed of hydrogen molecules at $0^{\circ} {C}$. (In ${m}/{s}$)View Solution
- 7When one mole of monatomic gas is mixed with one mole of a diatomic gas, then the equivalent value of $\gamma$ for the mixture will be (vibration mode neglected)View Solution
- 8View SolutionInternal energy of a non-ideal gas depends on ..........
- 9A gas at $ 27\ ^oC$ has a volume $V$ and pressure $P$. On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ....... $^oC$View Solution
- 10In Vander Waal’s equation $a$ and $b$ represent $\left( {P + \frac{a}{{{V^2}}}} \right)\,(V - b) = RT$View Solution
